In the realm of chemistry, ions are fundamental particles that play a crucial role in various natural phenomena and technological applications. From the electrolytes in our bodies to the batteries powering our devices, ions are everywhere. But What Are Ions exactly? Let’s delve into the world of these charged particles to understand their nature and significance.
Defining Ions: Charged Particles Explained
At the heart of understanding ions is the concept of electric charge. Atoms, the basic building blocks of matter, are typically electrically neutral, possessing an equal number of positively charged protons and negatively charged electrons. However, when an atom gains or loses electrons, this balance is disrupted, leading to the formation of an ion.
An ion, therefore, is an atom or molecule that has acquired an electrical charge by either gaining or losing electrons. This gain or loss of electrons fundamentally changes the electrical properties of the atom or molecule, transforming it into a charged species capable of interacting with other charged particles. Think of it like this: an atom is like a balanced scale, but when you add or remove weights (electrons), it becomes unbalanced – and becomes an ion.
 Ion3
Ion3
Image: Illustration depicting various types of ions including sodium, potassium, chloride and hydrogen-carbonate ions, key components in biological systems.
Types of Ions: Cations and Anions
Ions are broadly classified into two main types based on the nature of their charge:
-
Cations: These are positively charged ions. Cations are formed when an atom loses one or more electrons. By losing negatively charged electrons, the atom ends up with more protons than electrons, resulting in a net positive charge. Common examples of cations include sodium ions (Na+), potassium ions (K+), and calcium ions (Ca2+).
-
Anions: These are negatively charged ions. Anions are formed when an atom gains one or more electrons. By gaining negatively charged electrons, the atom ends up with more electrons than protons, resulting in a net negative charge. Common examples of anions include chloride ions (Cl-) and sulfate ions (SO42-).
The charge of an ion is determined by the number of electrons gained or lost. This is referred to as valency. We use terms like monovalent, divalent, and trivalent to describe ions with charges of +1 or -1, +2 or -2, and +3 or -3 respectively. For instance, Na+ is a monovalent cation, Cl- is a monovalent anion, Ca2+ is a divalent cation, and SO42- is a divalent anion.
Ions in Aqueous Solutions: Why Salt Conducts Electricity
Water, despite being a stable molecule (H2O), has a slight tendency to dissociate into hydrogen ions (H+) and hydroxide ions (OH-). This property allows water to dissolve many substances. However, the way substances interact with water and their behavior in aqueous solutions can vary significantly.
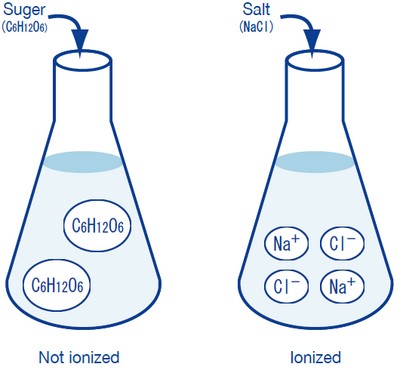
Consider the difference between sugar and salt when dissolved in water. Sugar (C6H12O6), a covalent compound, dissolves in water but remains as neutral molecules. It does not break down into ions. As a result, a sugar solution conducts electricity very poorly, similar to pure water.
On the other hand, salt (NaCl), an ionic compound, readily dissociates into sodium ions (Na+) and chloride ions (Cl-) when dissolved in water. These ions are mobile and carry electric charge. Therefore, a salt solution is a good conductor of electricity. This difference in electrical conductivity highlights the fundamental difference between ionic and covalent compounds in aqueous solutions. Ionic compounds like salt are formed through the electrostatic attraction between positively charged cations and negatively charged anions.
The Role of Ions in Our Bodies: Electrolytes and Physiology
Ions are not just confined to chemistry labs; they are essential for life itself. In biology, ions like sodium (Na+), potassium (K+), chloride (Cl-), and bicarbonate (HCO3-) are critical electrolytes found in bodily fluids such as blood and lymph.
These electrolytes play vital physiological roles, including:
- Maintaining Osmotic Pressure: Ions help regulate the balance of fluids inside and outside cells through osmotic pressure.
- Nerve and Muscle Function: Sodium, potassium, and calcium ions are crucial for nerve impulse transmission and muscle contraction.
- pH Balance: Bicarbonate ions are important in maintaining the pH balance of blood.
In medicine, electrolyte imbalances can indicate various health issues, highlighting the importance of ions in maintaining overall physiological well-being.
In Conclusion
Ions are charged particles formed from atoms or molecules gaining or losing electrons. They are categorized as cations (positive) and anions (negative) and are fundamental to understanding chemical reactions, electrical conductivity in solutions, and vital biological processes. From the salt in our food to the electrolytes in our bodies, understanding what are ions provides a crucial foundation for grasping the world around us at a molecular level.
Related Reading
The Basis of Ion