Are you curious about the building blocks of matter and how they interact? WHAT.EDU.VN can help. The valence electrons of an atom dictate its chemical behavior and how it forms bonds with other atoms. Understanding these outer shell electrons unlocks the secrets to chemical reactions, molecular structures, and the properties of materials. Explore concepts like electron configuration and the octet rule.
1. Understanding Valence Electrons: The Key to Chemical Bonding
Valence electrons are the electrons located in the outermost shell, or energy level, of an atom. These electrons are the primary actors in chemical bonding, determining how atoms interact to form molecules and compounds. The number and arrangement of valence electrons dictate an element’s chemical properties, reactivity, and the types of bonds it can form. Understanding valence electrons is fundamental to comprehending chemical behavior.
1.1. Defining Valence Electrons
Valence electrons are those electrons in the outermost electron shell of an atom. This shell is also known as the valence shell. These electrons are significant because they are involved in chemical bonding. The number of valence electrons an atom has determines how it will interact with other atoms. Atoms with a full valence shell are generally inert, while those with incomplete valence shells tend to form bonds to achieve stability.
1.2. The Significance of Valence Electrons
Why are valence electrons so important? Because they determine the chemical properties of an element. The number of valence electrons dictates how an atom will interact with other atoms, including whether it will form chemical bonds and what types of bonds it will form. This is crucial for understanding the behavior of elements in chemical reactions and the formation of molecules.
1.3. Valence vs Core Electrons
It’s important to distinguish between valence electrons and core electrons. Core electrons are those found in the inner shells of an atom, closer to the nucleus. These electrons are not typically involved in chemical bonding. Valence electrons, being in the outermost shell, are the ones that participate in bonding and determine the chemical behavior of the atom.
2. Determining the Number of Valence Electrons
So, how do you figure out how many valence electrons an atom has? One of the easiest methods is to use the periodic table. The group number (vertical column) of an element often indicates the number of valence electrons it possesses. This rule applies particularly well to the main group elements (groups 1, 2, and 13-18).
2.1. Using the Periodic Table
The periodic table is an invaluable tool for determining the number of valence electrons. Here’s how to use it:
- Groups 1 and 2: Elements in these groups (alkali and alkaline earth metals) have 1 and 2 valence electrons, respectively.
- Groups 13-18: For these groups, subtract 10 from the group number to find the number of valence electrons. For example, Group 16 (chalcogens) elements have 6 valence electrons.
- Transition Metals: The rule above doesn’t apply to transition metals (groups 3-12). The number of valence electrons in transition metals can vary due to their complex electron configurations.
2.2. Exceptions to the Rule
While the periodic table is a great guide, there are some exceptions, particularly among transition metals and heavier elements. These elements often have variable valencies, meaning they can exhibit different numbers of valence electrons depending on the chemical environment.
2.3. Electron Configuration and Valence Electrons
Another way to determine the number of valence electrons is by examining the electron configuration of an atom. The electron configuration describes the arrangement of electrons within the atom’s energy levels and sublevels. By identifying the outermost energy level, you can count the number of electrons in that level to find the number of valence electrons.
3. The Octet Rule and Chemical Stability
The octet rule is a guiding principle in understanding chemical bonding. It states that atoms tend to gain, lose, or share electrons to achieve a full valence shell with eight electrons, resembling the electron configuration of noble gases. Atoms that satisfy the octet rule are generally more stable and less reactive.
3.1. What is the Octet Rule?
The octet rule is based on the observation that atoms with eight valence electrons (an “octet”) are exceptionally stable. This stability arises from the filled s and p orbitals in the outermost shell. Atoms strive to achieve this stable configuration through chemical bonding.
3.2. How Atoms Achieve an Octet
Atoms can achieve an octet in several ways:
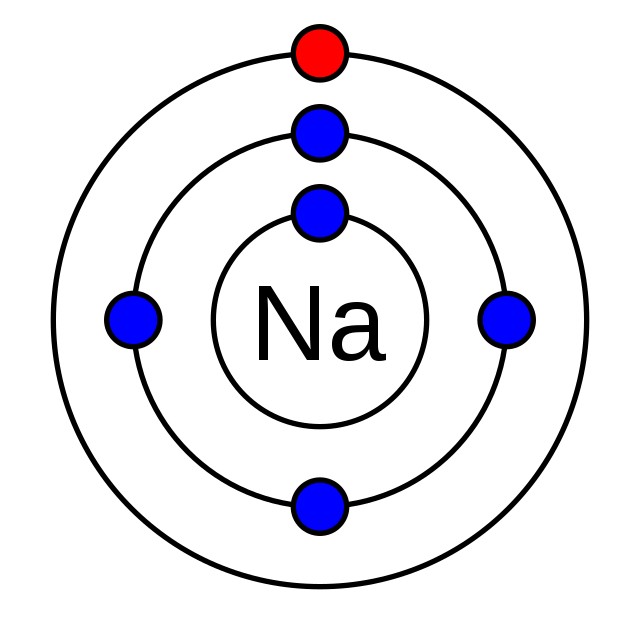
- Ionic Bonding: Atoms transfer electrons to form ions. For example, sodium (Na) donates an electron to chlorine (Cl), forming Na+ and Cl- ions, both with stable octets.
- Covalent Bonding: Atoms share electrons to achieve an octet. For example, carbon (C) shares electrons with four hydrogen (H) atoms to form methane (CH4), where each atom has a stable electron configuration.
- Metallic Bonding: Metal atoms share electrons in a “sea” of electrons, allowing each atom to achieve a stable electron configuration collectively.
3.3. Exceptions to the Octet Rule
While the octet rule is a useful guideline, it’s not universally applicable. Some exceptions include:
- Hydrogen (H): Only needs two electrons to fill its valence shell.
- Beryllium (Be): Often stable with four valence electrons.
- Boron (B): Can be stable with six valence electrons.
- Expanded Octets: Elements in the third period and beyond can sometimes accommodate more than eight electrons in their valence shells due to the availability of d orbitals.
4. Types of Chemical Bonds
Valence electrons play a central role in the formation of different types of chemical bonds: ionic bonds, covalent bonds, and metallic bonds. Each type of bond arises from the way valence electrons are distributed and shared between atoms.
4.1. Ionic Bonds
Ionic bonds form when one or more electrons are transferred from one atom to another, creating ions. These ions are oppositely charged and attract each other, forming a strong electrostatic attraction. Ionic bonds typically occur between metals and nonmetals.
4.2. Covalent Bonds
Covalent bonds form when atoms share one or more pairs of electrons. This sharing allows both atoms to achieve a stable electron configuration. Covalent bonds typically occur between nonmetals and can be single, double, or triple bonds, depending on the number of electron pairs shared.
4.3. Metallic Bonds
Metallic bonds occur in metals, where valence electrons are delocalized and shared among many atoms in a “sea” of electrons. This delocalization allows metals to conduct electricity and heat efficiently and gives them properties like malleability and ductility.
5. Valence Electrons and Chemical Reactions
The reactivity of an element is directly related to its valence electron configuration. Elements with incomplete valence shells are more reactive because they readily form bonds to achieve stability.
5.1. Reactivity of Elements
Elements with few or many valence electrons are generally more reactive. For example:
- Alkali Metals (Group 1): Highly reactive because they easily lose one electron to achieve a stable configuration.
- Halogens (Group 17): Highly reactive because they easily gain one electron to achieve a stable configuration.
- Noble Gases (Group 18): Inert (unreactive) because they already have a full valence shell.
5.2. Oxidation and Reduction
Oxidation and reduction (redox) reactions involve the transfer of electrons between atoms or molecules. The number of valence electrons an atom can gain, lose, or share determines its role in these reactions.
5.3. Catalysis and Valence Electrons
Catalysts are substances that speed up chemical reactions without being consumed in the process. Valence electrons play a key role in catalytic mechanisms, facilitating the formation and breaking of chemical bonds.
6. Advanced Concepts: Molecular Orbital Theory
For a deeper understanding of valence electrons and bonding, molecular orbital (MO) theory provides a more sophisticated model. MO theory explains how atomic orbitals combine to form molecular orbitals, which describe the distribution of electrons in a molecule.
6.1. Formation of Molecular Orbitals
Molecular orbitals are formed by the combination of atomic orbitals. When atoms bond, their atomic orbitals overlap to create new orbitals that span the entire molecule. These molecular orbitals can be bonding (lower energy) or antibonding (higher energy).
6.2. Bonding and Antibonding Orbitals
Bonding orbitals increase the stability of the molecule by concentrating electron density between the nuclei. Antibonding orbitals decrease the stability by having a node (zero electron density) between the nuclei.
6.3. Applications of Molecular Orbital Theory
MO theory is used to explain various chemical phenomena, including:
- Bond Order: The number of bonds between atoms in a molecule.
- Magnetic Properties: Whether a molecule is paramagnetic (attracted to a magnetic field) or diamagnetic (repelled by a magnetic field).
- Spectroscopic Properties: How molecules interact with light and other forms of electromagnetic radiation.
7. Practical Applications of Valence Electron Knowledge
Understanding valence electrons is not just theoretical; it has numerous practical applications in various fields.
7.1. Materials Science
In materials science, knowledge of valence electrons helps in the design of new materials with specific properties. For example, understanding how valence electrons behave in semiconductors is crucial for developing electronic devices.
7.2. Drug Discovery
In drug discovery, valence electrons are important for understanding how drugs interact with biological molecules. The way valence electrons are arranged in a drug molecule can determine its binding affinity to a target protein.
7.3. Environmental Science
In environmental science, valence electrons are important for understanding the behavior of pollutants in the environment. The way valence electrons are arranged in a pollutant molecule can determine its toxicity and how it interacts with other substances.
8. Common Questions About Valence Electrons
Here are some frequently asked questions about valence electrons, designed to clarify common points of confusion.
| Question | Answer |
|---|---|
| What Are Valence Electrons? | Valence electrons are the electrons in the outermost shell (valence shell) of an atom, responsible for chemical bonding. |
| How do I determine the number of valence electrons? | Use the periodic table group number (for main group elements) or examine the electron configuration. |
| What is the octet rule? | The octet rule states that atoms tend to gain, lose, or share electrons to achieve a full valence shell with eight electrons. |
| Are there exceptions to the octet rule? | Yes, exceptions include hydrogen (2 electrons), beryllium (4 electrons), boron (6 electrons), and elements that can have expanded octets. |
| What types of chemical bonds are formed by valence electrons? | Ionic bonds (transfer of electrons), covalent bonds (sharing of electrons), and metallic bonds (delocalization of electrons). |
| How do valence electrons affect the reactivity of an element? | Elements with incomplete valence shells are more reactive as they readily form bonds to achieve stability. |
| What is molecular orbital theory? | A theory explaining how atomic orbitals combine to form molecular orbitals, describing the distribution of electrons in a molecule. |
| Why are valence electrons important in materials science? | Understanding valence electrons helps in designing new materials with specific properties, like semiconductors. |
| How are valence electrons used in drug discovery? | The arrangement of valence electrons in a drug molecule determines its binding affinity to a target protein. |
| What role do valence electrons play in oxidation-reduction reactions? | Valence electrons are transferred between atoms or molecules in redox reactions, determining the oxidation states of the reacting species. |
9. Real-World Examples of Valence Electron Behavior
To further illustrate the importance of valence electrons, let’s consider a few real-world examples.
9.1. Sodium Chloride (Table Salt)
Sodium (Na) has one valence electron, while chlorine (Cl) has seven. Sodium readily donates its valence electron to chlorine, forming Na+ and Cl- ions. The electrostatic attraction between these ions results in the formation of sodium chloride (NaCl), common table salt.
9.2. Water (H2O)
Oxygen (O) has six valence electrons, while hydrogen (H) has one. Oxygen shares electrons with two hydrogen atoms, forming two covalent bonds. This sharing allows all atoms to achieve a stable electron configuration, resulting in the formation of water (H2O).
9.3. Diamond (C)
Carbon (C) has four valence electrons. In diamond, each carbon atom forms covalent bonds with four other carbon atoms, creating a strong, three-dimensional network. This network gives diamond its exceptional hardness and high melting point.
10. How to Learn More About Valence Electrons
If you’re eager to expand your knowledge of valence electrons and related topics, here are some resources:
- Textbooks: General chemistry textbooks provide comprehensive coverage of valence electrons and chemical bonding.
- Online Courses: Platforms like Coursera, edX, and Khan Academy offer courses on chemistry fundamentals.
- Scientific Journals: Publications like the Journal of the American Chemical Society and Angewandte Chemie publish cutting-edge research on chemical bonding and materials science.
- Educational Websites: Sites like Chemistry LibreTexts and HyperPhysics offer detailed explanations and interactive tools.
11. Importance of Valence Electrons in the Modern World
The understanding of valence electrons isn’t just an academic exercise; it’s vital for numerous technological advancements and everyday applications.
11.1. Development of New Technologies
The knowledge of valence electrons is crucial in the development of new technologies. For example, understanding how electrons behave in semiconductors is key to creating advanced electronic devices, such as smartphones, computers, and solar panels.
11.2. Medical Advancements
In the medical field, valence electrons play a role in designing new drugs and therapies. The interaction of drug molecules with biological targets is determined by the arrangement and behavior of valence electrons.
11.3. Sustainable Energy Solutions
The development of sustainable energy solutions, like solar cells and advanced batteries, relies on understanding and manipulating valence electrons in materials. This knowledge helps in creating more efficient and environmentally friendly energy technologies.
12. Advanced Topics Related to Valence Electrons
As you dive deeper into the world of valence electrons, you’ll encounter more advanced concepts that build upon the fundamentals.
12.1. Hybridization
Hybridization is the mixing of atomic orbitals to form new hybrid orbitals suitable for bonding. This concept explains the shapes and orientations of molecules. For example, carbon’s sp3 hybridization explains the tetrahedral geometry of methane (CH4).
12.2. Resonance
Resonance occurs when a molecule can be represented by multiple Lewis structures. The true structure is a hybrid of these resonance structures, with electrons delocalized over multiple bonds. This concept explains the stability and properties of molecules like benzene.
12.3. Ligand Field Theory
Ligand field theory describes the bonding and electronic structure of coordination complexes, where metal ions are surrounded by ligands (molecules or ions). This theory explains the colors and magnetic properties of transition metal complexes.
13. Continuing Your Education in Chemistry
To further your education in chemistry and deepen your understanding of valence electrons, consider the following steps:
- Take Advanced Courses: Enroll in advanced chemistry courses at a college or university.
- Conduct Research: Participate in research projects to gain hands-on experience in the lab.
- Join Professional Organizations: Become a member of organizations like the American Chemical Society to network with other chemists and stay updated on the latest research.
14. Expert Insights on Valence Electrons
According to Dr. Linus Pauling, a renowned chemist and Nobel laureate, “The nature of the chemical bond is determined by the arrangement and behavior of valence electrons.” This highlights the fundamental importance of valence electrons in understanding chemical phenomena.
15. The Role of WHAT.EDU.VN in Answering Your Chemistry Questions
If you’re still puzzled about valence electrons or any other chemistry topic, don’t hesitate to reach out to WHAT.EDU.VN. Our platform provides a space to ask questions and receive clear, concise answers from experts. We are here to make learning chemistry easier and more accessible for everyone.
16. Beyond the Basics: Advanced Bonding Theories
To gain a more complete understanding of how atoms interact, it’s important to explore advanced bonding theories.
16.1. Density Functional Theory (DFT)
Density Functional Theory (DFT) is a computational method used to investigate the electronic structure of atoms, molecules, and solids. DFT calculates the electronic properties based on the electron density, providing accurate predictions for molecular properties and chemical reactions.
16.2. Valence Bond Theory
Valence Bond (VB) Theory is an alternative to Molecular Orbital (MO) Theory. It describes chemical bonds as localized interactions between atoms, focusing on the overlap of atomic orbitals to form bonds. VB Theory is particularly useful for understanding resonance and the localized nature of chemical bonds.
17. Addressing Common Misconceptions About Valence Electrons
It’s common to encounter misconceptions when learning about valence electrons. Let’s address some of the most frequent misunderstandings.
17.1. Misconception: Valence Electrons Are Only Important in Covalent Bonds
Clarification: While valence electrons are crucial in covalent bonds, they are also involved in ionic and metallic bonds. In ionic bonds, valence electrons are transferred, leading to the formation of ions. In metallic bonds, valence electrons are delocalized, creating a sea of electrons.
17.2. Misconception: All Atoms Strive to Achieve an Octet
Clarification: While many atoms follow the octet rule, there are exceptions. Hydrogen needs only two electrons, and some elements can have expanded octets.
17.3. Misconception: Valence Electrons Are Identical to All Electrons in the Outermost Shell
Clarification: Valence electrons are specifically those in the outermost shell that participate in bonding. Inner electrons are not considered valence electrons.
18. The Future of Valence Electron Research
Research on valence electrons continues to drive advancements in various fields. Future directions include:
18.1. Quantum Computing
Understanding the behavior of valence electrons is essential for developing quantum computing technologies. Quantum computers rely on the precise control of electron states, and valence electrons play a key role in these systems.
18.2. Nanomaterials
The design and synthesis of nanomaterials require a deep understanding of valence electrons. The properties of nanomaterials are highly dependent on the electronic structure of their constituent atoms.
18.3. Green Chemistry
Research on valence electrons is contributing to the development of green chemistry practices, which aim to minimize the environmental impact of chemical processes.
19. Need Help? Contact WHAT.EDU.VN Today
Still have questions about valence electrons? Our team at WHAT.EDU.VN is here to help. We provide a platform for asking any question and receiving expert answers. Contact us today to get the support you need.
Address: 888 Question City Plaza, Seattle, WA 98101, United States
WhatsApp: +1 (206) 555-7890
Website: WHAT.EDU.VN
20. How WHAT.EDU.VN Makes Learning Easier
We understand that chemistry can be challenging, but with WHAT.EDU.VN, learning becomes easier and more accessible. Our platform provides a user-friendly environment where you can ask questions, receive detailed answers, and connect with experts.
20.1. Instant Answers
Get quick and accurate answers to your questions.
20.2. Expert Explanations
Receive clear and concise explanations from knowledgeable professionals.
20.3. Free Access
Enjoy free access to our vast knowledge base and support services.
21. Interactive Tools for Understanding Valence Electrons
To aid in your learning, consider using interactive tools that visually represent valence electrons and chemical bonding.
21.1. Online Simulations
Use online simulations to visualize electron configurations and bonding processes. These simulations can help you understand how valence electrons interact in different chemical environments.
21.2. Molecular Modeling Software
Explore molecular modeling software to build and manipulate molecules, observing how valence electrons influence their structure and properties.
21.3. Virtual Reality (VR) Chemistry Labs
Immerse yourself in virtual reality chemistry labs to conduct experiments and explore chemical concepts in a realistic and engaging environment.
22. Exploring the History of Valence Electron Theory
Understanding the history of valence electron theory provides valuable context and insights into how our current understanding evolved.
22.1. Early Concepts
The concept of valence was first introduced in the mid-19th century by chemists like August Kekulé and Archibald Scott Couper, who recognized that atoms have a fixed capacity for bonding.
22.2. Gilbert N. Lewis and the Octet Rule
In the early 20th century, Gilbert N. Lewis developed the octet rule and introduced the concept of electron sharing in covalent bonds, revolutionizing our understanding of chemical bonding.
22.3. Linus Pauling and Quantum Mechanics
Linus Pauling applied quantum mechanics to chemical bonding, developing the concept of hybridization and further refining our understanding of valence electrons and molecular structure.
23. Understanding Electronegativity and Valence Electrons
Electronegativity is a measure of an atom’s ability to attract electrons in a chemical bond. It’s closely related to valence electrons and helps predict the type of bond that will form between atoms.
23.1. Electronegativity Trends
Electronegativity generally increases across the periodic table from left to right and decreases from top to bottom. These trends are influenced by the number of valence electrons and the distance of these electrons from the nucleus.
23.2. Predicting Bond Polarity
The difference in electronegativity between two atoms can predict the polarity of a bond. A large difference indicates an ionic bond, while a small difference indicates a covalent bond.
24. Valence Electrons and Coordination Chemistry
Coordination chemistry involves the study of coordination complexes, where metal ions are surrounded by ligands. Valence electrons play a crucial role in the formation and properties of these complexes.
24.1. Ligand Donation
Ligands donate electron pairs to the metal ion, forming coordinate covalent bonds. The number and type of ligands determine the geometry and electronic properties of the complex.
24.2. Crystal Field Theory
Crystal field theory explains how the electronic structure of the metal ion is affected by the surrounding ligands, influencing the color and magnetic properties of the complex.
25. The Importance of Hands-On Experience
While theoretical knowledge is essential, hands-on experience can greatly enhance your understanding of valence electrons and chemical bonding.
25.1. Laboratory Experiments
Conducting laboratory experiments allows you to observe chemical reactions firsthand and apply your knowledge of valence electrons to real-world scenarios.
25.2. Model Building
Building molecular models can help you visualize the three-dimensional structures of molecules and understand how valence electrons influence their shapes.
26. How to Ask Effective Questions on WHAT.EDU.VN
To get the best answers on WHAT.EDU.VN, follow these tips for asking effective questions:
26.1. Be Specific
Provide as much detail as possible about your question, including any relevant background information.
26.2. Use Clear Language
Use clear and concise language to ensure that your question is easy to understand.
26.3. Provide Context
Explain what you already know about the topic and what you are trying to learn.
27. Ethical Considerations in Chemistry Research
As you advance in your study of chemistry, it’s important to consider ethical issues.
27.1. Responsible Conduct of Research
Adhere to the highest standards of integrity and honesty in your research.
27.2. Safety in the Lab
Follow all safety protocols and guidelines to ensure a safe working environment.
28. Staying Current with Chemistry News and Research
To stay informed about the latest developments in chemistry, follow reputable news sources and research journals.
28.1. Scientific Journals
Read articles in journals such as the Journal of the American Chemical Society, Nature Chemistry, and Angewandte Chemie.
28.2. Science News Websites
Follow science news websites such as ScienceDaily, Phys.org, and EurekAlert!
29. Encouraging a Love for Chemistry
Finally, remember that learning chemistry should be an enjoyable and rewarding experience.
29.1. Find Connections to Real Life
Look for ways to connect chemistry to your everyday life, such as understanding the chemistry of cooking or the science behind cosmetics.
29.2. Share Your Knowledge
Share your knowledge with others and help them appreciate the beauty and importance of chemistry.
Valence electrons are the foundation of chemical bonding and reactivity. Whether you’re a student, a curious learner, or a professional, understanding valence electrons is essential for comprehending the world around you. And remember, WHAT.EDU.VN is here to support your learning journey.
Still have questions? Don’t hesitate to ask! Our team is ready to provide you with expert answers and clear explanations. Contact us today at what.edu.vn, and let us help you unlock the secrets of chemistry.
