What Is A Chemical Compound? It’s a substance formed when two or more chemical elements chemically bond together. At WHAT.EDU.VN, we provide clear explanations and examples to help you understand chemical compounds and their importance in our world, offering a solution to your questions. Delve into the world of chemistry and learn about atomic combinations, molecular structures, and chemical elements.
1. Defining Chemical Compounds: The Building Blocks of Matter
A chemical compound is a substance composed of two or more different elements chemically bonded together in a fixed ratio. These elements are held together by chemical bonds, such as ionic or covalent bonds, forming a stable molecule or crystal lattice. Understanding the nature and formation of chemical compounds is fundamental to grasping the properties of matter.
1.1. What Distinguishes a Compound from an Element or a Mixture?
-
Element: An element is a pure substance consisting of only one type of atom, such as gold (Au) or oxygen (O).
-
Compound: A compound is formed when two or more different elements chemically combine in a fixed ratio, like water (H2O) or sodium chloride (NaCl).
-
Mixture: A mixture is a combination of two or more substances that are physically combined but not chemically bonded. Mixtures can be separated by physical means, such as filtration or evaporation. An example is saltwater, a mixture of water (H2O) and sodium chloride (NaCl).
1.2. Why Are Chemical Compounds Important?
Chemical compounds are essential because they form the basis of all matter around us. They determine the properties of substances and their interactions. From the air we breathe (a mixture containing oxygen, nitrogen, and other compounds) to the food we eat (composed of various organic and inorganic compounds), chemical compounds play a crucial role in our daily lives. At WHAT.EDU.VN, we believe understanding these fundamental building blocks allows individuals to grasp the complex interactions that govern our world.
1.3. Where Can I Learn More About Chemical Compounds?
For those eager to explore the fascinating world of chemical compounds, WHAT.EDU.VN is your go-to resource. Whether you’re a student tackling chemistry homework, a curious adult eager to expand your knowledge, or anyone in between, our platform provides a wealth of easy-to-understand information. Plus, if you have burning questions, our free Q&A service connects you with experts ready to provide clear and concise answers. Learn about atomic combinations, molecular structures, and the myriad of ways elements combine to create the world around us.
2. Formation of Chemical Compounds: Bonding and Interactions
The formation of chemical compounds involves the interaction of atoms through chemical bonds. These bonds arise from the electromagnetic forces between atoms, leading to the formation of stable molecules or crystal lattices. Understanding the different types of chemical bonds is crucial for understanding how compounds are formed and their properties.
2.1. What Are the Main Types of Chemical Bonds?
-
Ionic Bonds: Ionic bonds occur when electrons are transferred from one atom to another, creating ions (charged atoms). These oppositely charged ions are attracted to each other, forming an ionic compound. A classic example is sodium chloride (NaCl), where sodium (Na) loses an electron to chlorine (Cl), forming Na+ and Cl- ions.
-
Covalent Bonds: Covalent bonds involve the sharing of electrons between atoms. These bonds typically occur between nonmetal atoms. Depending on how equally the electrons are shared, the bond can be polar (unequal sharing) or nonpolar (equal sharing). Water (H2O) is an example of a polar covalent compound, while methane (CH4) is an example of a nonpolar covalent compound.
-
Metallic Bonds: Metallic bonds are found in metals and involve the sharing of electrons among a lattice of metal atoms. These “sea” of electrons allows metals to conduct electricity and heat efficiently and gives them their characteristic properties such as malleability and ductility.
2.2. What Role Does Electronegativity Play?
Electronegativity is a measure of an atom’s ability to attract electrons in a chemical bond. The difference in electronegativity between two atoms determines the type of bond that will form. A large difference in electronegativity (typically greater than 1.7) leads to ionic bonds, while a smaller difference results in covalent bonds.
2.3. How Does Molecular Geometry Affect Compound Properties?
The three-dimensional arrangement of atoms in a molecule, known as molecular geometry, significantly impacts the compound’s physical and chemical properties. Molecular geometry is determined by the repulsion between electron pairs around the central atom, as described by the Valence Shell Electron Pair Repulsion (VSEPR) theory. For example, the bent shape of a water molecule (H2O) makes it polar, while the tetrahedral shape of methane (CH4) makes it nonpolar.
2.4. Where Can I Get Clear Explanations on Chemical Bonding?
Navigating the complexities of chemical bonding can feel daunting. At WHAT.EDU.VN, we break down these concepts into digestible pieces, perfect for learners of all levels. Our platform offers a comprehensive overview of the different types of chemical bonds, the role of electronegativity, and the impact of molecular geometry on compound properties. Plus, if you find yourself stuck on a tricky problem, our free Q&A service is there to help. Don’t hesitate to ask your questions and receive expert guidance to deepen your understanding of chemical compounds and their formation.
3. Types of Chemical Compounds: Organic vs. Inorganic
Chemical compounds are broadly classified into two main categories: organic and inorganic. Organic compounds are primarily composed of carbon and hydrogen atoms, while inorganic compounds include all other compounds. This classification is based on historical reasons and the unique properties of carbon.
3.1. What Characterizes Organic Compounds?
Organic compounds typically contain carbon-hydrogen (C-H) bonds and often include other elements such as oxygen, nitrogen, and halogens. They are the building blocks of living organisms and play a central role in biology and biochemistry. The ability of carbon to form long chains and rings leads to a vast diversity of organic compounds.
3.2. What Distinguishes Inorganic Compounds?
Inorganic compounds encompass all compounds that are not organic. They include a wide variety of substances such as salts, metals, minerals, and coordination complexes. Inorganic compounds play essential roles in geology, materials science, and industrial chemistry.
3.3. What Are Some Common Examples of Each Type?
-
Organic Compounds: Methane (CH4), ethanol (C2H5OH), glucose (C6H12O6), and benzene (C6H6) are common examples of organic compounds.
-
Inorganic Compounds: Sodium chloride (NaCl), water (H2O), carbon dioxide (CO2), and ammonia (NH3) are common examples of inorganic compounds.
3.4. Why is the Distinction Between Organic and Inorganic Important?
The distinction between organic and inorganic compounds is crucial due to their different properties and applications. Organic compounds are often flammable, have lower melting and boiling points, and are soluble in organic solvents. Inorganic compounds tend to have higher melting and boiling points, are soluble in water, and are often used in structural materials and industrial processes.
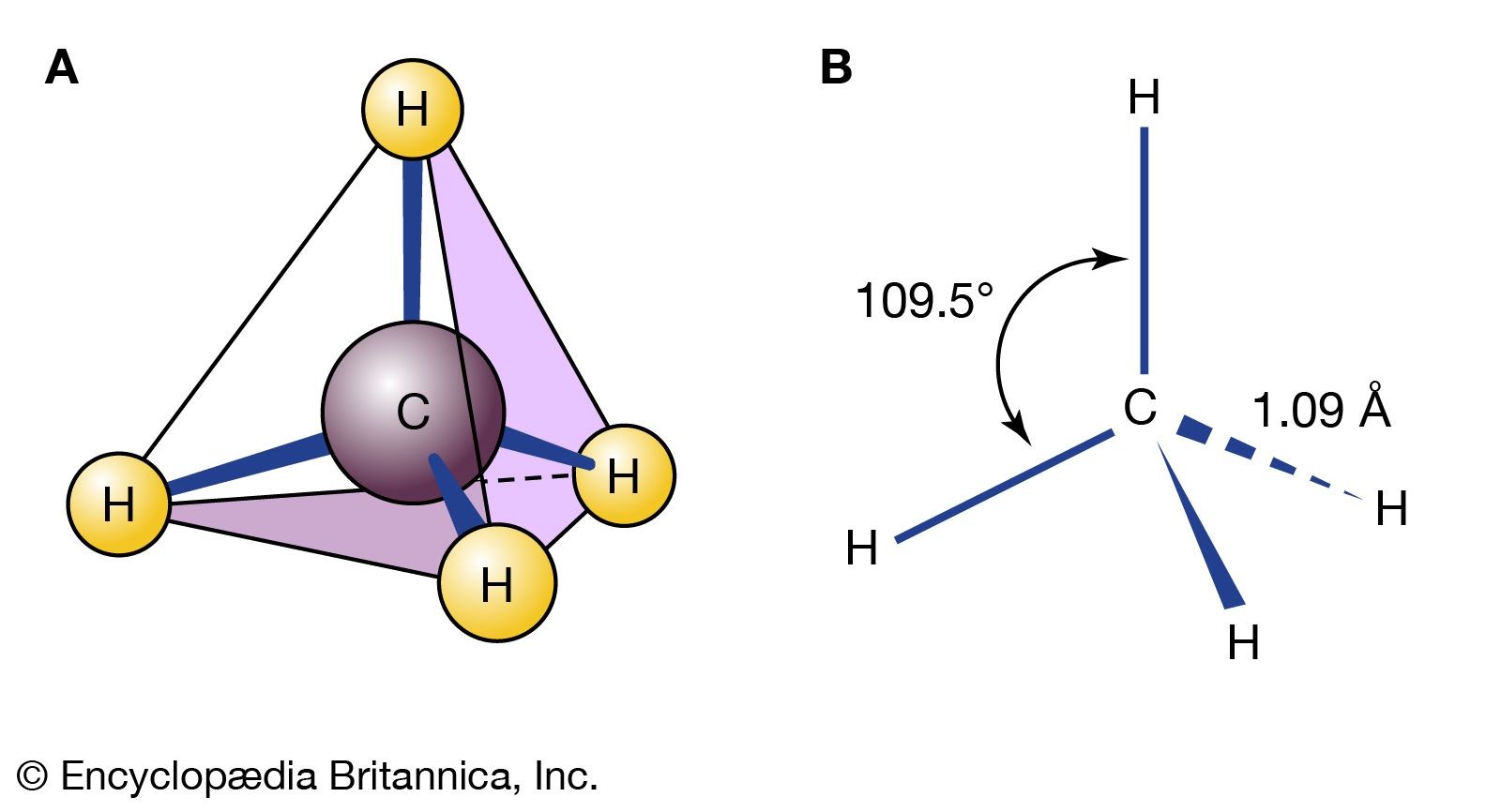 methane molecule
methane molecule
3.5. Where Can I Find Clear Explanations and Examples of Organic and Inorganic Compounds?
Understanding the difference between organic and inorganic compounds is essential for anyone studying chemistry. At WHAT.EDU.VN, we offer a wealth of resources to help you master this topic. Our platform provides clear definitions, examples, and explanations of the unique properties and applications of each type of compound. Plus, if you have specific questions or need help with a challenging assignment, our free Q&A service is there to assist you. Ask your questions, and our experts will provide personalized guidance to deepen your understanding.
4. Properties of Chemical Compounds: Physical and Chemical
Chemical compounds exhibit a wide range of physical and chemical properties that are determined by their composition, structure, and bonding. These properties dictate how compounds behave in different environments and how they interact with other substances.
4.1. What Are Physical Properties?
Physical properties are characteristics that can be observed or measured without changing the chemical identity of the substance. Examples include:
- Melting Point: The temperature at which a solid turns into a liquid.
- Boiling Point: The temperature at which a liquid turns into a gas.
- Density: The mass per unit volume of a substance.
- Solubility: The ability of a substance to dissolve in a solvent.
- Color: The visual appearance of a substance.
- Odor: The scent of a substance.
4.2. What Are Chemical Properties?
Chemical properties describe how a substance reacts with other substances or changes its chemical composition. Examples include:
- Flammability: The ability of a substance to burn.
- Reactivity: The tendency of a substance to undergo chemical reactions.
- Acidity/Basicity: The ability of a substance to donate or accept protons (H+).
- Oxidation/Reduction Potential: The tendency of a substance to gain or lose electrons.
4.3. How Do Intermolecular Forces Influence Physical Properties?
Intermolecular forces (IMFs) are the attractive or repulsive forces between molecules. These forces significantly affect the physical properties of compounds, such as melting point, boiling point, and solubility. Stronger IMFs lead to higher melting and boiling points, while weaker IMFs result in lower melting and boiling points.
4.4. What Role Does Molecular Structure Play in Chemical Reactivity?
The molecular structure of a compound determines its chemical reactivity. The arrangement of atoms and bonds within a molecule influences how it interacts with other substances. For example, molecules with weaker bonds or reactive functional groups tend to be more reactive.
4.5. Where Can I Explore the Intricate World of Chemical Compound Properties?
Delving into the properties of chemical compounds can unlock a deeper understanding of the world around us. At WHAT.EDU.VN, we provide a comprehensive exploration of physical and chemical properties, explaining how they are determined by composition, structure, and bonding. Our platform covers everything from melting and boiling points to flammability and reactivity. Plus, if you have specific questions or need clarification on a particular concept, our free Q&A service is available to help. Ask your questions and receive expert guidance to deepen your understanding of chemical compounds and their properties.
5. Nomenclature of Chemical Compounds: Naming Conventions
The nomenclature of chemical compounds involves systematic naming conventions that allow chemists to identify and communicate about substances clearly. These conventions are governed by the International Union of Pure and Applied Chemistry (IUPAC).
5.1. What Are the Basic Rules for Naming Ionic Compounds?
Ionic compounds are typically named by combining the name of the cation (positive ion) followed by the name of the anion (negative ion). For example, NaCl is named sodium chloride. If the cation has multiple possible charges, a Roman numeral is used to indicate the charge, such as iron(II) chloride (FeCl2) and iron(III) chloride (FeCl3).
5.2. What Are the Basic Rules for Naming Covalent Compounds?
Covalent compounds are named using prefixes to indicate the number of atoms of each element in the molecule. For example, CO2 is named carbon dioxide, and N2O4 is named dinitrogen tetroxide.
5.3. How Are Organic Compounds Named?
The nomenclature of organic compounds follows a more complex set of rules based on the longest continuous carbon chain, the presence of functional groups, and the position of substituents. For example, CH3CH2OH is named ethanol, and CH3COOH is named acetic acid.
5.4. What Are Common Exceptions to the Naming Rules?
Some common compounds have trivial names that are widely used instead of their systematic names. Examples include water (H2O), ammonia (NH3), and methane (CH4).
5.5. How Can WHAT.EDU.VN Help Me Master the Nomenclature of Chemical Compounds?
Chemical nomenclature can seem like a foreign language at first. But with the right guidance, you can confidently name and identify chemical compounds. At WHAT.EDU.VN, we offer clear, step-by-step instructions on how to name ionic, covalent, and organic compounds. Our platform provides numerous examples and practice exercises to reinforce your understanding. And if you encounter any challenges, our free Q&A service is there to help. Ask your questions and receive expert assistance to master chemical nomenclature.
6. Chemical Reactions and Equations: Compound Transformations
Chemical reactions involve the transformation of chemical compounds into new substances. These transformations are represented by chemical equations, which show the reactants (starting materials) and products (resulting substances). Understanding chemical reactions and equations is essential for studying chemistry.
6.1. What Are the Main Types of Chemical Reactions?
- Synthesis Reactions: Two or more reactants combine to form a single product (A + B → AB).
- Decomposition Reactions: A single reactant breaks down into two or more products (AB → A + B).
- Single Displacement Reactions: One element replaces another in a compound (A + BC → AC + B).
- Double Displacement Reactions: Two compounds exchange ions to form two new compounds (AB + CD → AD + CB).
- Combustion Reactions: A substance reacts with oxygen, producing heat and light (CxHy + O2 → CO2 + H2O).
6.2. How Are Chemical Equations Balanced?
Chemical equations must be balanced to satisfy the law of conservation of mass, which states that matter cannot be created or destroyed in a chemical reaction. Balancing equations involves adjusting the coefficients (numbers in front of the chemical formulas) so that the number of atoms of each element is the same on both sides of the equation.
6.3. What Information Do Chemical Equations Provide?
Chemical equations provide information about the reactants, products, and stoichiometry (relative amounts) of the substances involved in a chemical reaction. They also indicate the physical states of the substances (solid, liquid, gas, or aqueous solution) and the reaction conditions (temperature, pressure, catalyst).
6.4. What Are Common Indicators of a Chemical Reaction?
- Color Change: A change in the color of the reaction mixture.
- Formation of a Precipitate: A solid that forms from a solution.
- Gas Evolution: The production of bubbles.
- Heat Release or Absorption: An increase or decrease in temperature.
6.5. Where Can I Deepen My Understanding of Chemical Reactions and Equations?
Mastering chemical reactions and equations is a cornerstone of chemistry education. At WHAT.EDU.VN, we offer comprehensive resources to help you succeed in this area. Our platform provides detailed explanations of the different types of chemical reactions, step-by-step instructions on balancing equations, and clear guidance on interpreting chemical equations. Plus, if you have questions or need assistance with a specific problem, our free Q&A service is available to help. Ask your questions and receive expert guidance to master chemical reactions and equations.
7. Acids, Bases, and Salts: Essential Chemical Compounds
Acids, bases, and salts are essential classes of chemical compounds that play crucial roles in chemistry and biology. They are characterized by their distinct properties and reactions in aqueous solutions.
7.1. What Defines an Acid?
Acids are substances that donate protons (H+) or accept electrons. They have a sour taste, turn litmus paper red, and react with bases to form salts and water. Common examples include hydrochloric acid (HCl), sulfuric acid (H2SO4), and acetic acid (CH3COOH).
7.2. What Defines a Base?
Bases are substances that accept protons (H+) or donate electrons. They have a bitter taste, turn litmus paper blue, and react with acids to form salts and water. Common examples include sodium hydroxide (NaOH), potassium hydroxide (KOH), and ammonia (NH3).
7.3. What Are Salts?
Salts are ionic compounds formed from the reaction of an acid and a base. They consist of a cation (positive ion) and an anion (negative ion). Common examples include sodium chloride (NaCl), potassium nitrate (KNO3), and calcium carbonate (CaCO3).
7.4. What Is the pH Scale?
The pH scale is a measure of the acidity or basicity of a solution. It ranges from 0 to 14, with 7 being neutral, values less than 7 being acidic, and values greater than 7 being basic. The pH is defined as the negative logarithm of the hydrogen ion concentration ([H+]).
7.5. How Does WHAT.EDU.VN Simplify the Study of Acids, Bases, and Salts?
Understanding acids, bases, and salts is essential for anyone studying chemistry. At WHAT.EDU.VN, we offer clear, concise explanations of these important chemical compounds, along with examples and practical applications. Our platform covers everything from acid-base reactions to pH calculations. Plus, if you have questions or need help with a challenging assignment, our free Q&A service is there to assist you. Ask your questions and receive expert guidance to master acids, bases, and salts.
8. Chemical Compounds in Everyday Life: Applications and Uses
Chemical compounds are ubiquitous in our daily lives, playing essential roles in various applications and industries. From the food we eat to the medicines we take, chemical compounds are integral to modern society.
8.1. What Are Some Common Chemical Compounds Found in Food?
- Water (H2O): Essential for hydration and many biological processes.
- Sodium Chloride (NaCl): Table salt, used for flavoring and preservation.
- Glucose (C6H12O6): A sugar that provides energy.
- Acetic Acid (CH3COOH): Vinegar, used as a preservative and flavoring agent.
8.2. What Are Some Common Chemical Compounds Found in Medicines?
- Aspirin (C9H8O4): Used as a pain reliever and anti-inflammatory.
- Paracetamol (C8H9NO2): Used as a pain reliever and fever reducer.
- Amoxicillin (C16H19N3O5S): An antibiotic used to treat bacterial infections.
8.3. What Are Some Common Chemical Compounds Used in Household Products?
- Sodium Hypochlorite (NaClO): Bleach, used for cleaning and disinfection.
- Sodium Carbonate (Na2CO3): Washing soda, used as a cleaning agent.
- Ammonia (NH3): Used in cleaning products and fertilizers.
8.4. How Are Chemical Compounds Used in Industry?
Chemical compounds are used in a wide range of industrial processes, including:
- Polymer Production: Polymers such as polyethylene and polypropylene are used to make plastics.
- Fertilizer Production: Ammonia and nitrates are used to make fertilizers for agriculture.
- Pharmaceutical Production: Various chemical compounds are used to synthesize drugs and medicines.
8.5. How Can WHAT.EDU.VN Help Me Explore the Applications of Chemical Compounds in Everyday Life?
Understanding the role of chemical compounds in our daily lives can make chemistry more relevant and engaging. At WHAT.EDU.VN, we offer a wealth of information on the applications of chemical compounds in food, medicine, household products, and industry. Our platform provides real-world examples and clear explanations to help you connect chemistry to your everyday experiences. Plus, if you have questions or want to learn more about a particular compound, our free Q&A service is there to assist you. Ask your questions and receive expert guidance to deepen your understanding.
9. Safety Considerations When Working With Chemical Compounds
Working with chemical compounds requires careful attention to safety to prevent accidents and health hazards. Understanding the potential risks and implementing appropriate safety measures is essential for protecting yourself and others.
9.1. What Are Common Hazards Associated With Chemical Compounds?
- Toxicity: Some compounds can be harmful or deadly if ingested, inhaled, or absorbed through the skin.
- Flammability: Some compounds can easily catch fire.
- Corrosivity: Some compounds can damage or destroy materials, including skin and eyes.
- Reactivity: Some compounds can react violently with other substances.
9.2. What Safety Equipment Should Be Used When Handling Chemicals?
- Safety Goggles: To protect your eyes from splashes and fumes.
- Gloves: To protect your hands from skin contact with chemicals.
- Lab Coat: To protect your clothing from spills.
- Fume Hood: To prevent inhalation of hazardous vapors.
9.3. What Are Basic Safety Rules for Handling Chemical Compounds?
- Read Labels: Always read and understand the labels on chemical containers.
- Follow Instructions: Follow instructions carefully when performing experiments or handling chemicals.
- Avoid Mixing Chemicals: Never mix chemicals unless explicitly instructed to do so.
- Proper Disposal: Dispose of chemical waste properly according to safety guidelines.
9.4. What Should You Do in Case of a Chemical Spill or Accident?
- Alert Others: Immediately inform others in the area about the spill or accident.
- Contain the Spill: If possible, contain the spill to prevent it from spreading.
- Clean Up: Clean up the spill according to safety guidelines.
- Seek Medical Attention: If you are injured or exposed to a hazardous chemical, seek medical attention immediately.
9.5. Where Can I Find Comprehensive Safety Information for Working With Chemicals?
Ensuring your safety when working with chemical compounds is paramount. At WHAT.EDU.VN, we provide comprehensive safety information, including common hazards, necessary safety equipment, basic safety rules, and procedures for handling spills or accidents. Our platform emphasizes the importance of understanding potential risks and implementing appropriate safety measures. Plus, if you have specific safety-related questions or need clarification on a particular chemical, our free Q&A service is there to assist you. Ask your questions and receive expert guidance to ensure a safe working environment.
10. Recent Advances in Chemical Compound Research
The field of chemical compound research is constantly evolving, with new discoveries and innovations occurring regularly. These advances have significant implications for various fields, including medicine, materials science, and environmental science.
10.1. What Are Some Recent Discoveries in Drug Development?
- New Antibiotics: Researchers are developing new antibiotics to combat antibiotic-resistant bacteria.
- Cancer Therapies: New chemical compounds are being developed for targeted cancer therapies.
- Neurodegenerative Disease Treatments: Researchers are exploring chemical compounds to treat Alzheimer’s and Parkinson’s diseases.
10.2. What Are Some Recent Advances in Materials Science?
- Graphene: This two-dimensional carbon material has unique properties and potential applications in electronics and energy storage.
- Perovskites: These materials are being developed for solar cells and other energy applications.
- Shape-Memory Alloys: These materials can return to their original shape after being deformed.
10.3. What Are Some Recent Innovations in Environmental Science?
- Carbon Capture: Chemical compounds are being developed to capture carbon dioxide from the atmosphere.
- Water Purification: New materials are being developed to remove pollutants from water.
- Biodegradable Plastics: Researchers are exploring biodegradable polymers to reduce plastic waste.
10.4. How Are Chemical Compounds Being Used to Address Global Challenges?
Chemical compounds are playing a crucial role in addressing global challenges such as climate change, disease, and resource scarcity. Innovations in chemical compound research are leading to new solutions for these pressing issues.
10.5. How Can WHAT.EDU.VN Keep Me Updated on the Latest Advances in Chemical Compound Research?
Staying informed about the latest advances in chemical compound research is essential for anyone interested in science and technology. At WHAT.EDU.VN, we provide timely updates on new discoveries, innovations, and applications in this dynamic field. Our platform covers a wide range of topics, including drug development, materials science, and environmental science. Plus, if you have questions about a particular area of research or want to learn more about a specific chemical compound, our free Q&A service is there to assist you. Ask your questions and receive expert guidance to stay at the forefront of chemical compound research.
Do you have more questions about chemical compounds or any other topic? Don’t hesitate to ask us at WHAT.EDU.VN! Our team of experts is ready to provide you with clear, concise answers and help you expand your knowledge. Visit us at 888 Question City Plaza, Seattle, WA 98101, United States, or contact us via WhatsApp at +1 (206) 555-7890. We are here to help you learn and grow. Explore the world with what.edu.vn!