Polymers are a class of substances, both natural and synthetic, that are fundamental to our world. From the plastics in everyday items to the biological macromolecules essential for life, polymers are everywhere. These substances are characterized by their large molecules, known as macromolecules, which are essentially long chains made up of repeating smaller chemical units called monomers. Understanding what polymers are, their types, and their significance is crucial in various fields of science and engineering.
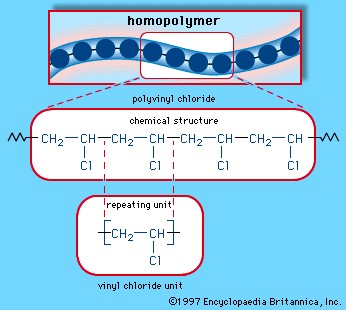
Polymers, at their core, are giant molecules formed through a process called polymerization, where many monomers link together. This linkage can create chains, networks, and complex structures depending on the type of monomers and the reaction conditions. While the term “polymer” implies many units, when the number of monomer units is exceptionally large, it’s often termed a high polymer. It’s important to note that polymers aren’t limited to being made from a single type of monomer. They can be composed of identical monomers, known as homopolymers, or a combination of different monomers, known as copolymers. This versatility in composition allows for a vast array of polymers with diverse properties.
Polymers are broadly categorized into natural and synthetic types. Natural polymers occur in nature and are vital for biological systems. Organic natural polymers are crucial for life. For instance, plants are largely composed of polymers such as cellulose, lignin, and resins. Cellulose, a polysaccharide, is made of sugar molecules and provides structural support to plants. Lignin is a complex polymer network contributing to the rigidity of plant cell walls. Rubber, another familiar organic polymer, is a polymer of isoprene.
Proteins, essential for countless biological functions, are polymers of amino acids. Nucleic acids, including DNA and RNA, which carry genetic information, are polymers of nucleotides. Starches, a primary energy source from plants, are also natural polymers composed of glucose. These examples highlight the indispensable role of organic polymers in living organisms.
Beyond organic polymers, inorganic natural polymers also exist. Diamond and graphite, both forms of carbon, are examples. Diamond’s hardness arises from its three-dimensional network of carbon atoms, while graphite’s layered structure allows it to be used as a lubricant.
Synthetic polymers, on the other hand, are man-made and form the basis of many industrial and consumer products. Polyvinyl chloride (PVC), as illustrated earlier, is a synthetic homopolymer made from repeating vinyl chloride units and widely used in construction and various products. Other synthetic polymers include polyethylene, polypropylene, nylon, and polyester, each tailored for specific applications due to their unique properties. These materials have revolutionized industries, offering properties like flexibility, durability, and cost-effectiveness.
In conclusion, polymers are ubiquitous and vital substances. Whether natural or synthetic, organic or inorganic, they are the building blocks of countless materials and life itself. From the strength of diamonds to the elasticity of rubber and the versatility of plastics, understanding polymers is key to unlocking further innovations in science, technology, and beyond.