Organic compound: Are you curious to know what they are? At WHAT.EDU.VN, we offer a clear definition of organic compounds, explore their diverse types, and uncover their essential uses. If you’re looking for quick, free answers, you’re in the right place. Let’s explore carbon compounds, organic molecules and chemical structures.
1. Understanding Organic Compounds
Organic compounds are a vast and diverse class of chemical compounds that contain carbon atoms covalently linked to other atoms, typically hydrogen, oxygen, or nitrogen. These compounds form the building blocks of life and are essential to various aspects of our daily lives.
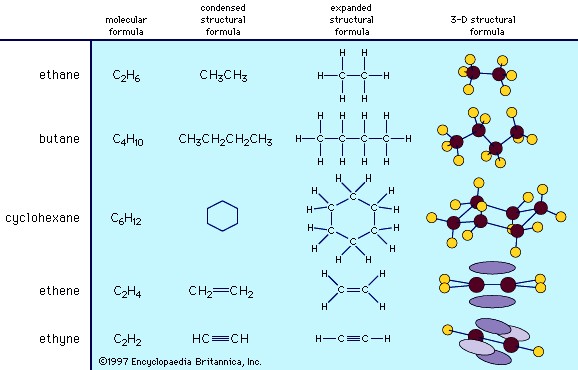 structural formulas of some organic compounds
structural formulas of some organic compounds
2. Key Characteristics of Organic Compounds
Organic compounds possess several key characteristics that distinguish them from inorganic compounds:
- Carbon Backbone: The defining feature of organic compounds is the presence of a carbon backbone. Carbon atoms have the unique ability to form stable covalent bonds with each other, creating long chains or rings that serve as the structural framework for organic molecules.
- Covalent Bonding: Organic compounds primarily consist of covalent bonds, where atoms share electrons to form stable molecules. This type of bonding is essential for the diversity and complexity of organic structures.
- Presence of Hydrogen: Most organic compounds contain hydrogen atoms bonded to carbon atoms. These carbon-hydrogen bonds contribute to the overall stability and reactivity of organic molecules.
- Other Elements: In addition to carbon and hydrogen, organic compounds may also contain other elements such as oxygen, nitrogen, sulfur, phosphorus, and halogens. These elements introduce functional groups that influence the chemical properties and reactivity of the molecule.
- Isomerism: Organic compounds can exhibit isomerism, where molecules have the same molecular formula but different structural arrangements. This phenomenon leads to a vast number of unique organic compounds with distinct properties.
3. Historical Perspective on Organic Compounds
3.1. The Vital Force Theory
Initially, scientists believed that organic compounds could only be produced by living organisms due to a “vital force” inherent in living matter. This theory hindered the understanding and synthesis of organic compounds.
3.2. Wöhler’s Breakthrough
In 1828, Friedrich Wöhler accidentally synthesized urea, an organic compound, from inorganic materials. This groundbreaking discovery challenged the vital force theory and paved the way for the development of modern organic chemistry.
3.3. Modern Understanding
Today, we understand that organic compounds are governed by the same chemical principles as inorganic compounds. The unique properties of organic compounds arise from the specific arrangement and bonding of carbon atoms and other elements.
4. Carbon Bonding: The Foundation of Organic Chemistry
4.1. Carbon’s Unique Bonding Ability
The carbon atom’s unique ability to form extensive networks of covalent bonds with itself and other elements is the cornerstone of organic chemistry. This bonding versatility allows for the creation of an immense variety of organic molecules.
4.2. Tetrahedral Geometry
When fully bonded, the four bonds of a carbon atom are directed towards the corners of a tetrahedron, forming angles of approximately 109.5 degrees. This three-dimensional arrangement contributes to the diversity of organic structures.
4.3. Single, Double, and Triple Bonds
Carbon atoms can form single, double, or triple bonds with each other, further expanding the possibilities for structural variation. These multiple bonds have distinct electronic properties that influence the reactivity of organic molecules.
5. Functional Groups: The Reactive Sites of Organic Molecules
5.1. Definition and Importance
Functional groups are specific atoms or groups of atoms within a molecule that are responsible for its characteristic chemical reactions. These groups confer specific reactivity patterns on the molecules they are part of, making them essential for understanding organic chemistry.
5.2. Common Functional Groups
Some common functional groups include:
- Alkanes: Compounds containing only carbon and hydrogen atoms linked by single bonds. They are relatively unreactive and form the inert framework of many organic compounds.
- Alkenes: Compounds containing a carbon-carbon double bond. The pi bond in the double bond makes alkenes more reactive than alkanes.
- Alkynes: Compounds containing a carbon-carbon triple bond. Alkynes are even more reactive than alkenes due to the presence of two pi bonds.
- Aromatic Hydrocarbons (Arenes): Compounds containing a benzene ring, a six-carbon ring with alternating single and double bonds. Aromatic compounds have unique stability and reactivity.
- Alcohols: Compounds containing a hydroxyl (-OH) group bonded to a carbon atom. Alcohols are versatile compounds that can participate in a variety of reactions.
- Ethers: Compounds containing an oxygen atom bonded to two carbon atoms. Ethers are relatively unreactive and are often used as solvents.
- Thiols: Compounds containing a sulfur atom bonded to a hydrogen atom (-SH). Thiols have a strong, unpleasant odor and are important in biological systems.
- Amines: Compounds containing a nitrogen atom bonded to hydrogen atoms or alkyl/aryl groups. Amines are basic compounds that can act as nucleophiles.
- Halides: Compounds containing a halogen atom (fluorine, chlorine, bromine, or iodine) bonded to a carbon atom. Halides are versatile building blocks for organic synthesis.
- Aldehydes: Compounds containing a carbonyl group (C=O) with at least one hydrogen atom bonded to the carbonyl carbon. Aldehydes are reactive compounds that can be easily oxidized.
- Ketones: Compounds containing a carbonyl group (C=O) with two carbon atoms bonded to the carbonyl carbon. Ketones are less reactive than aldehydes but are still important in organic chemistry.
- Carboxylic Acids: Compounds containing a carboxyl group (-COOH), which is a combination of a carbonyl group and a hydroxyl group. Carboxylic acids are acidic compounds that can donate a proton.
5.3. Influence on Reactivity
The presence of specific functional groups dictates the reactivity of an organic molecule. Functional groups with polar bonds or multiple bonds are more reactive than those with only nonpolar single bonds.
6. Types of Organic Compounds
6.1. Hydrocarbons
Hydrocarbons are organic compounds composed solely of carbon and hydrogen atoms. They are the simplest type of organic compound and serve as the foundation for many other organic molecules.
6.1.1. Alkanes
Alkanes are saturated hydrocarbons containing only single bonds. They are relatively unreactive and are used as fuels and lubricants.
6.1.2. Alkenes
Alkenes are unsaturated hydrocarbons containing at least one carbon-carbon double bond. They are more reactive than alkanes and are used in the production of plastics and other chemicals.
6.1.3. Alkynes
Alkynes are unsaturated hydrocarbons containing at least one carbon-carbon triple bond. They are highly reactive and are used in welding and the synthesis of complex organic molecules.
6.1.4. Aromatic Hydrocarbons
Aromatic hydrocarbons contain a benzene ring, a six-carbon ring with alternating single and double bonds. They have unique stability and are used in the production of pharmaceuticals, dyes, and plastics.
6.2. Alcohols
Alcohols are organic compounds containing a hydroxyl (-OH) group bonded to a carbon atom. They are versatile compounds used as solvents, disinfectants, and in the production of beverages and pharmaceuticals.
6.3. Ethers
Ethers are organic compounds containing an oxygen atom bonded to two carbon atoms. They are relatively unreactive and are used as solvents and anesthetics.
6.4. Amines
Amines are organic compounds containing a nitrogen atom bonded to hydrogen atoms or alkyl/aryl groups. They are basic compounds used in the production of dyes, pharmaceuticals, and polymers.
6.5. Aldehydes and Ketones
Aldehydes and ketones are organic compounds containing a carbonyl group (C=O). Aldehydes have at least one hydrogen atom bonded to the carbonyl carbon, while ketones have two carbon atoms bonded to the carbonyl carbon. They are used as flavorings, fragrances, and in the production of polymers.
6.6. Carboxylic Acids
Carboxylic acids are organic compounds containing a carboxyl group (-COOH). They are acidic compounds used in the production of soaps, detergents, and pharmaceuticals.
6.7. Esters
Esters are organic compounds formed by the reaction of a carboxylic acid and an alcohol. They have pleasant odors and are used as flavorings, fragrances, and solvents.
6.8. Amides
Amides are organic compounds containing a nitrogen atom bonded to a carbonyl group. They are important in biological systems, particularly in proteins and peptides.
7. Importance of Organic Compounds
7.1. Building Blocks of Life
Organic compounds are essential for all known life forms. They make up the structures of cells, tissues, and organs, and they participate in the chemical reactions that sustain life.
7.2. Food and Nutrition
Carbohydrates, proteins, and fats, the three major classes of food, are all organic compounds. They provide energy, build and repair tissues, and regulate bodily functions.
7.3. Medicine and Pharmaceuticals
Many drugs and pharmaceuticals are organic compounds designed to interact with specific biological targets in the body. They are used to treat a wide range of diseases and conditions.
7.4. Materials and Manufacturing
Organic compounds are used in the production of plastics, polymers, fibers, and other materials that are essential for modern manufacturing.
7.5. Energy and Fuels
Fossil fuels, such as petroleum, natural gas, and coal, are composed of organic compounds. They are used as a primary source of energy for transportation, electricity generation, and heating.
8. Examples of Organic Compounds
8.1. Methane (CH4)
Methane is the simplest organic compound, a colorless and odorless gas that is the main component of natural gas.
8.2. Ethanol (C2H5OH)
Ethanol is an alcohol used as a solvent, disinfectant, and in the production of alcoholic beverages.
8.3. Acetic Acid (CH3COOH)
Acetic acid is a carboxylic acid found in vinegar, giving it its characteristic sour taste.
8.4. Glucose (C6H12O6)
Glucose is a simple sugar (carbohydrate) that is a primary source of energy for living organisms.
8.5. Benzene (C6H6)
Benzene is an aromatic hydrocarbon used in the production of plastics, resins, and synthetic fibers.
9. Common Reactions of Organic Compounds
9.1. Addition Reactions
Addition reactions involve the addition of atoms or groups of atoms to a molecule, typically at a double or triple bond.
9.2. Substitution Reactions
Substitution reactions involve the replacement of one atom or group of atoms in a molecule with another atom or group of atoms.
9.3. Elimination Reactions
Elimination reactions involve the removal of atoms or groups of atoms from a molecule, typically forming a double or triple bond.
9.4. Oxidation-Reduction Reactions
Oxidation-reduction (redox) reactions involve the transfer of electrons between molecules. Oxidation is the loss of electrons, while reduction is the gain of electrons.
10. Nomenclature of Organic Compounds
10.1. IUPAC Nomenclature
The International Union of Pure and Applied Chemistry (IUPAC) provides a standardized system for naming organic compounds. This system ensures that each compound has a unique and unambiguous name.
10.2. Common Names
In addition to IUPAC names, some organic compounds are known by common names that have historical or traditional origins.
11. Applications of Organic Compounds
11.1. Polymers and Plastics
Polymers are large molecules made up of repeating structural units called monomers. Many plastics are polymers derived from organic compounds.
11.2. Pharmaceuticals
Many drugs and pharmaceuticals are organic compounds designed to interact with specific biological targets in the body.
11.3. Agrochemicals
Agrochemicals, such as pesticides and herbicides, are organic compounds used to protect crops from pests and diseases.
11.4. Dyes and Pigments
Dyes and pigments are organic compounds used to color textiles, paints, and other materials.
11.5. Solvents
Many organic compounds are used as solvents to dissolve other substances.
12. Environmental Impact of Organic Compounds
12.1. Pollution
Some organic compounds can be pollutants that contaminate air, water, and soil.
12.2. Toxicity
Some organic compounds are toxic and can have harmful effects on human health and the environment.
12.3. Sustainability
Sustainable chemistry seeks to develop environmentally friendly processes for the production and use of organic compounds.
13. Recent Advances in Organic Chemistry
13.1. Green Chemistry
Green chemistry focuses on designing chemical processes that minimize or eliminate the use and generation of hazardous substances.
13.2. Catalysis
Catalysis involves the use of catalysts to speed up chemical reactions and improve their efficiency.
13.3. Supramolecular Chemistry
Supramolecular chemistry studies the interactions between molecules and the formation of complex structures.
13.4. Nanotechnology
Nanotechnology involves the manipulation of matter at the nanoscale, including the design and synthesis of organic nanomaterials.
14. Resources for Further Learning
14.1. Textbooks
- Organic Chemistry by Paula Yurkanis Bruice
- Organic Chemistry by Kenneth L. Williamson
14.2. Online Courses
- Coursera: Organic Chemistry
- edX: Introduction to Organic Chemistry
14.3. Websites
- Khan Academy: Organic Chemistry
- Chemistry LibreTexts: Organic Chemistry
15. Importance of Asking Questions
Learning about organic compounds can be challenging, and asking questions is a crucial part of the learning process. Don’t hesitate to seek clarification and explore concepts further.
15.1. The Value of Curiosity
Curiosity drives exploration and discovery. Asking questions fuels your understanding and helps you connect new information to what you already know.
15.2. Overcoming Challenges
Organic chemistry can be complex, but asking questions allows you to break down difficult topics into manageable parts.
15.3. Deepening Understanding
Asking questions encourages critical thinking and helps you develop a deeper understanding of organic compounds and their properties.
16. Organic Compounds in Everyday Life
16.1. Household Products
Many household products, such as cleaning agents, detergents, and cosmetics, contain organic compounds.
16.2. Clothing and Textiles
Natural fibers like cotton and wool, as well as synthetic fibers like polyester and nylon, are made of organic compounds.
16.3. Plastics and Packaging
Plastics used in packaging, containers, and various household items are derived from organic polymers.
16.4. Fuels and Energy
Gasoline, diesel, and natural gas are organic compounds used as fuels for transportation and energy production.
17. The Future of Organic Chemistry
17.1. Sustainable Materials
Researchers are developing sustainable organic materials that are biodegradable and environmentally friendly.
17.2. Personalized Medicine
Organic chemistry plays a key role in the development of personalized medicine, where treatments are tailored to an individual’s genetic makeup.
17.3. Advanced Electronics
Organic electronics, such as OLEDs (organic light-emitting diodes), are revolutionizing display technology and lighting.
17.4. Renewable Energy
Organic compounds are being explored for use in solar cells and other renewable energy technologies.
18. Exploring Complex Organic Structures
18.1. Proteins
Proteins are complex organic molecules made up of amino acids. They perform a wide range of functions in living organisms, including catalyzing reactions, transporting molecules, and providing structural support.
18.2. Nucleic Acids
Nucleic acids, such as DNA and RNA, are complex organic molecules that carry genetic information. They are essential for heredity and protein synthesis.
18.3. Lipids
Lipids, such as fats, oils, and waxes, are organic molecules that are insoluble in water. They serve as energy storage, insulation, and structural components of cell membranes.
18.4. Carbohydrates
Carbohydrates, such as sugars and starches, are organic molecules that provide energy and structural support.
19. Safety Precautions When Handling Organic Compounds
19.1. Use of Personal Protective Equipment (PPE)
Always wear appropriate PPE, such as gloves, goggles, and lab coats, when handling organic compounds.
19.2. Proper Ventilation
Work in a well-ventilated area to avoid inhaling toxic vapors.
19.3. Safe Disposal Methods
Dispose of organic compounds properly according to established safety protocols.
19.4. Emergency Procedures
Know the location of safety equipment and emergency procedures in case of spills or accidents.
20. Careers in Organic Chemistry
20.1. Research Scientist
Research scientists conduct experiments and develop new organic compounds and materials.
20.2. Pharmaceutical Chemist
Pharmaceutical chemists design and synthesize new drugs and pharmaceuticals.
20.3. Chemical Engineer
Chemical engineers apply the principles of chemistry to design and operate chemical processes.
20.4. Environmental Chemist
Environmental chemists study the impact of chemicals on the environment and develop methods for pollution control.
21. The Role of Organic Chemistry in Drug Development
21.1. Target Identification
Organic chemists identify and validate biological targets for drug development.
21.2. Lead Discovery
Organic chemists design and synthesize lead compounds that interact with the target.
21.3. Optimization
Organic chemists optimize the structure of lead compounds to improve their potency, selectivity, and pharmacokinetic properties.
21.4. Synthesis
Organic chemists develop efficient and scalable methods for synthesizing drug candidates.
22. The Importance of Understanding Chemical Structures
22.1. Predicting Properties
Understanding chemical structures allows chemists to predict the physical and chemical properties of organic compounds.
22.2. Designing Reactions
Chemists use their knowledge of chemical structures to design and carry out chemical reactions.
22.3. Interpreting Data
Chemical structures are essential for interpreting spectroscopic data and understanding reaction mechanisms.
22.4. Communicating Information
Chemical structures provide a clear and concise way to communicate information about organic compounds.
23. Common Misconceptions About Organic Compounds
23.1. “Organic” Means “Natural”
The term “organic” in chemistry refers to compounds containing carbon, not necessarily those derived from natural sources.
23.2. All Organic Compounds Are Safe
Some organic compounds are toxic and can have harmful effects on human health and the environment.
23.3. Organic Chemistry Is Too Difficult
While challenging, organic chemistry can be mastered with dedication and a solid understanding of basic concepts.
23.4. Organic Chemistry Is Only Relevant to Chemists
Organic chemistry is relevant to a wide range of fields, including medicine, materials science, and environmental science.
24. The Significance of Stereochemistry in Organic Chemistry
24.1. Chirality
Chirality refers to the property of a molecule that is non-superimposable on its mirror image.
24.2. Enantiomers
Enantiomers are stereoisomers that are mirror images of each other.
24.3. Diastereomers
Diastereomers are stereoisomers that are not mirror images of each other.
24.4. Importance in Biology
Stereochemistry is crucial in biology, as many biological molecules are chiral and interact selectively with specific stereoisomers.
25. Organic Chemistry and the Environment
25.1. Green Chemistry Principles
Green chemistry principles aim to reduce the environmental impact of chemical processes by minimizing waste, using safer chemicals, and increasing energy efficiency.
25.2. Sustainable Synthesis
Sustainable synthesis involves the development of chemical reactions that are environmentally friendly and use renewable resources.
25.3. Pollution Remediation
Organic chemistry plays a key role in developing methods for cleaning up pollution and remediating contaminated sites.
25.4. Biodegradable Materials
Researchers are developing biodegradable organic materials that can decompose naturally, reducing plastic waste.
26. The Role of Spectroscopy in Identifying Organic Compounds
26.1. Nuclear Magnetic Resonance (NMR) Spectroscopy
NMR spectroscopy provides information about the structure and environment of atoms in a molecule.
26.2. Infrared (IR) Spectroscopy
IR spectroscopy provides information about the functional groups present in a molecule.
26.3. Mass Spectrometry (MS)
Mass spectrometry provides information about the molecular weight and fragmentation pattern of a molecule.
26.4. UV-Vis Spectroscopy
UV-Vis spectroscopy provides information about the electronic transitions in a molecule.
27. Key Concepts in Organic Reaction Mechanisms
27.1. Nucleophiles and Electrophiles
Nucleophiles are electron-rich species that attack electron-deficient centers, while electrophiles are electron-deficient species that attack electron-rich centers.
27.2. Carbocations and Carbanions
Carbocations are positively charged carbon ions, while carbanions are negatively charged carbon ions.
27.3. Concerted and Stepwise Reactions
Concerted reactions occur in a single step, while stepwise reactions occur in multiple steps.
27.4. Transition States and Intermediates
Transition states are high-energy structures that represent the point of maximum energy during a reaction, while intermediates are stable species that exist during a reaction.
28. Challenges in Organic Synthesis
28.1. Selectivity
Achieving high selectivity in organic synthesis, where only the desired product is formed, can be challenging.
28.2. Yield
Maximizing the yield of a reaction, the amount of product obtained relative to the starting materials, is a key goal in organic synthesis.
28.3. Protecting Groups
Protecting groups are used to temporarily block the reactivity of a functional group during a reaction.
28.4. Stereocontrol
Controlling the stereochemistry of a reaction, the spatial arrangement of atoms in the product, can be difficult.
29. The Future of Organic Electronics
29.1. OLEDs (Organic Light-Emitting Diodes)
OLEDs are organic materials that emit light when an electric current is applied. They are used in displays and lighting.
29.2. Organic Solar Cells
Organic solar cells are devices that convert sunlight into electricity using organic materials.
29.3. Flexible Electronics
Organic materials can be used to create flexible and wearable electronic devices.
29.4. Sensors
Organic materials can be used to create sensors that detect specific chemicals or biological molecules.
30. FAQ about What Is An Organic Compound
| Question | Answer |
|---|---|
| What is the primary element found in all organic compounds? | Carbon is the primary element found in all organic compounds. |
| Are all compounds containing carbon classified as organic? | No, some carbon-containing compounds like carbon dioxide, carbonates, and carbides are classified as inorganic. |
| What types of bonds are common in organic compounds? | Covalent bonds are the most common type of bond in organic compounds, especially carbon-carbon and carbon-hydrogen bonds. |
| Why is carbon so important in organic chemistry? | Carbon’s ability to form stable bonds with itself and a variety of other elements allows for a vast diversity of organic molecules. |
| What is a functional group? | A functional group is a specific group of atoms within a molecule that is responsible for characteristic chemical reactions. |
| Can organic compounds be found in non-living things? | Yes, organic compounds can be found in non-living things like petroleum, natural gas, and plastics, even though they are primarily associated with living organisms. |
| What are hydrocarbons? | Hydrocarbons are organic compounds composed solely of carbon and hydrogen atoms. |
| How do organic compounds contribute to pharmaceuticals? | Many drugs and pharmaceuticals are organic compounds designed to interact with specific biological targets in the body, treating a wide range of diseases and conditions. |
| What role do organic compounds play in food? | Carbohydrates, proteins, and fats, which are essential nutrients in food, are all organic compounds that provide energy and support bodily functions. |
| What are some environmental concerns related to organic compounds? | Some organic compounds can be pollutants that contaminate air, water, and soil. Sustainable chemistry seeks to develop environmentally friendly processes for the production and use of organic compounds to minimize these impacts. |
Do you have more questions about organic compounds or anything else? Don’t struggle with unanswered questions. At WHAT.EDU.VN, we provide a platform where you can ask any question and receive free answers from experts and a knowledgeable community.
Address: 888 Question City Plaza, Seattle, WA 98101, United States
Whatsapp: +1 (206) 555-7890
Website: what.edu.vn
Submit your questions today and get the answers you need quickly and easily!

