Chemical weathering is the process that breaks down rocks by changing their chemical composition. Have questions about this fascinating natural phenomenon? At WHAT.EDU.VN, we make understanding complex topics easy and accessible. Get clear explanations, real-world examples, and expert insights into chemical decomposition, acid rain effects, and mineral alteration. Discover how chemical weathering shapes our world and find answers to all your questions at WHAT.EDU.VN.
1. Understanding Chemical Weathering
Chemical weathering involves the alteration of the chemical structure of rocks and minerals at or near the Earth’s surface. This process contrasts with physical weathering, which breaks rocks into smaller pieces without changing their chemical makeup. Instead, chemical weathering transforms the original materials into new substances through chemical reactions.
Chemical weathering is a crucial component of the Earth’s natural recycling system. It helps to break down rocks and minerals, releasing essential nutrients into the soil and water systems. These nutrients support plant growth and maintain ecological balance. The products of chemical weathering are also vital for forming sedimentary rocks and shaping landscapes.
1.1. Defining Chemical Weathering
Chemical weathering can be defined as the decomposition of rocks and minerals through chemical reactions. These reactions occur when water, oxygen, acids, and other chemical agents interact with rock materials. The result is the transformation of the original minerals into new compounds that are more stable under surface conditions.
The reactions involved in chemical weathering are influenced by several factors, including temperature, moisture, and the presence of reactive chemicals. For example, warmer temperatures can accelerate reaction rates, while the availability of water facilitates the transport of reactants and products.
1.2. The Role of Chemical Weathering in the Earth System
Chemical weathering plays a significant role in the Earth’s geochemical cycles. It helps regulate the composition of the atmosphere, oceans, and soils by controlling the release and uptake of various elements.
For instance, the weathering of silicate rocks consumes carbon dioxide ($CO_2$) from the atmosphere, reducing greenhouse gas levels and influencing global climate. The dissolved ions released during weathering are transported to the oceans, where they contribute to the formation of marine sediments and regulate ocean acidity.
In soils, chemical weathering provides essential nutrients for plant growth. The breakdown of minerals releases elements such as potassium, calcium, and phosphorus, which are vital for plant nutrition. These nutrients support terrestrial ecosystems and influence agricultural productivity.
2. Types of Chemical Weathering Processes
Several types of chemical weathering processes contribute to the breakdown of rocks and minerals. These processes include dissolution, hydrolysis, oxidation, carbonation, and biological weathering. Each process involves different chemical reactions and affects various types of rocks and minerals.
2.1. Dissolution
Dissolution is the process by which minerals dissolve in water. It is particularly effective on soluble minerals such as halite (sodium chloride) and gypsum (calcium sulfate). Water acts as a solvent, breaking the chemical bonds that hold the mineral together and dispersing the ions into the solution.
2.1.1. How Dissolution Works
The dissolution process begins when water molecules come into contact with the surface of a mineral. Water molecules are polar, meaning they have a slight positive charge on one side and a slight negative charge on the other. This polarity allows water to interact with the charged ions in the mineral structure.
For example, when water interacts with halite (NaCl), the positive hydrogen atoms in water are attracted to the negative chloride ions ($Cl^−$), while the negative oxygen atoms are attracted to the positive sodium ions ($Na^+$). This interaction weakens the ionic bonds in the halite crystal, causing it to break apart and dissolve into sodium and chloride ions.
2.1.2. Examples of Dissolution in Nature
-
Karst Topography: The dissolution of limestone ($CaCO_3$) by acidic groundwater creates distinctive karst landscapes characterized by caves, sinkholes, and underground drainage systems.
-
Salt Flats: The dissolution of salt deposits in arid regions leads to the formation of salt flats or playas, where dissolved salts precipitate as water evaporates.
-
Water Softening: Dissolution is also used in water softening processes to remove calcium and magnesium ions from hard water by dissolving them with chemicals.
2.2. Hydrolysis
Hydrolysis is a chemical reaction in which water reacts with a mineral, causing it to break down into new compounds. This process is particularly important in the weathering of silicate minerals, which are the primary components of many rocks.
2.2.1. The Hydrolysis Process Explained
In hydrolysis, water molecules react with the minerals, leading to the formation of new minerals and dissolved ions. The hydrogen ions ($H^+$) and hydroxide ions ($OH^−$) from water replace ions in the mineral structure, altering its composition and stability.
For example, the hydrolysis of feldspar minerals (such as orthoclase, $KAlSi_3O_8$) can produce clay minerals (such as kaolinite, $Al_2Si_2O_5(OH)_4$) and dissolved ions. The reaction can be represented as follows:
$2 KAlSi_3O_8(s) + 2 H_2O(l) + 2 H^+(aq) → Al_2Si_2O_5(OH)_4(s) + 2 K^+(aq) + 4 SiO_2(aq)$
In this reaction, orthoclase reacts with water and hydrogen ions to form kaolinite, potassium ions, and dissolved silica. The clay mineral kaolinite is more stable at the Earth’s surface than feldspar, making hydrolysis an essential weathering process.
2.2.2. Examples of Hydrolysis in Nature
-
Clay Formation: Hydrolysis is the primary process responsible for the formation of clay minerals in soils and sediments. Clay minerals play a crucial role in soil fertility, water retention, and contaminant transport.
-
Weathering of Granite: The hydrolysis of feldspar minerals in granite leads to the gradual breakdown of the rock, forming a sandy or gravelly residue known as grus.
-
Alteration of Volcanic Ash: Hydrolysis can rapidly alter volcanic ash deposits, transforming them into clay-rich soils that are fertile and productive.
2.3. Oxidation
Oxidation is the process by which minerals react with oxygen, causing them to lose electrons. This process is particularly important in the weathering of iron-rich minerals, such as pyrite ($FeS_2$) and olivine ($(Mg, Fe)_2SiO_4$).
2.3.1. Understanding Oxidation Reactions
In oxidation, oxygen atoms gain electrons from the mineral, leading to the formation of new compounds with a higher oxidation state. The most common example of oxidation is the rusting of iron, where iron atoms react with oxygen to form iron oxide (rust).
For example, the oxidation of pyrite ($FeS_2$) can produce iron oxide ($Fe_2O_3$) and sulfuric acid ($H_2SO_4$). The reaction can be represented as follows:
$4 FeS_2(s) + 15 O_2(g) + 14 H_2O(l) → 4 Fe_2O_3(s) + 8 H_2SO_4(aq)$
In this reaction, pyrite reacts with oxygen and water to form iron oxide (hematite) and sulfuric acid. The sulfuric acid can further contribute to chemical weathering by dissolving other minerals.
2.3.2. Examples of Oxidation in Nature
-
Rusting of Iron: The oxidation of iron-rich rocks and minerals leads to the formation of rust-colored coatings and stains on the rock surface.
-
Acid Mine Drainage: The oxidation of sulfide minerals in mine tailings can generate sulfuric acid, leading to acid mine drainage, which pollutes water and harms aquatic ecosystems.
-
Weathering of Basalt: The oxidation of iron-rich minerals in basalt can cause the rock to crumble and disintegrate, forming a reddish-brown soil.
2.4. Carbonation
Carbonation is the process by which minerals react with carbonic acid ($H_2CO_3$), which is formed when carbon dioxide ($CO_2$) dissolves in water. This process is particularly important in the weathering of carbonate rocks, such as limestone and chalk.
2.4.1. How Carbonation Works
When carbon dioxide dissolves in water, it forms carbonic acid, which can then react with carbonate minerals. The carbonic acid breaks down the carbonate minerals, forming calcium bicarbonate ($Ca(HCO_3)_2$), which is soluble in water and can be easily carried away.
The reaction can be represented as follows:
$CaCO_3(s) + H_2CO_3(aq) → Ca^{2+}(aq) + 2 HCO_3^−(aq)$
In this reaction, calcium carbonate (limestone) reacts with carbonic acid to form calcium ions and bicarbonate ions, which are dissolved in water.
2.4.2. Examples of Carbonation in Nature
-
Formation of Caves: The dissolution of limestone by carbonic acid creates caves and underground drainage systems in karst regions.
-
Weathering of Marble: The carbonation of marble statues and buildings can cause them to deteriorate over time, leading to the loss of intricate details and features.
-
Regulation of Ocean Acidity: The dissolution of carbonate minerals in the ocean helps to regulate ocean acidity by consuming excess carbon dioxide.
2.5. Biological Weathering
Biological weathering involves the breakdown of rocks and minerals by living organisms, such as plants, animals, and microbes. This process can occur through both physical and chemical means.
2.5.1. The Role of Living Organisms
-
Plants: Plant roots can exert physical pressure on rocks, causing them to crack and break apart. Additionally, plants can release organic acids that dissolve minerals and promote chemical weathering.
-
Animals: Burrowing animals, such as earthworms and rodents, can physically break down rocks and expose fresh surfaces to weathering. They also contribute to chemical weathering by mixing organic matter into the soil, which enhances the activity of microbes.
-
Microbes: Microorganisms, such as bacteria and fungi, can secrete organic acids and enzymes that dissolve minerals and promote chemical weathering. They also play a crucial role in the oxidation and reduction of minerals.
2.5.2. Examples of Biological Weathering in Nature
-
Lichen Weathering: Lichens can colonize rock surfaces and secrete organic acids that dissolve minerals, leading to the gradual breakdown of the rock.
-
Root Wedging: Tree roots can grow into cracks in rocks, exerting pressure that causes the rocks to split apart.
-
Microbial Weathering: Microbes can accelerate the weathering of rocks and minerals by producing organic acids and enzymes that dissolve minerals.
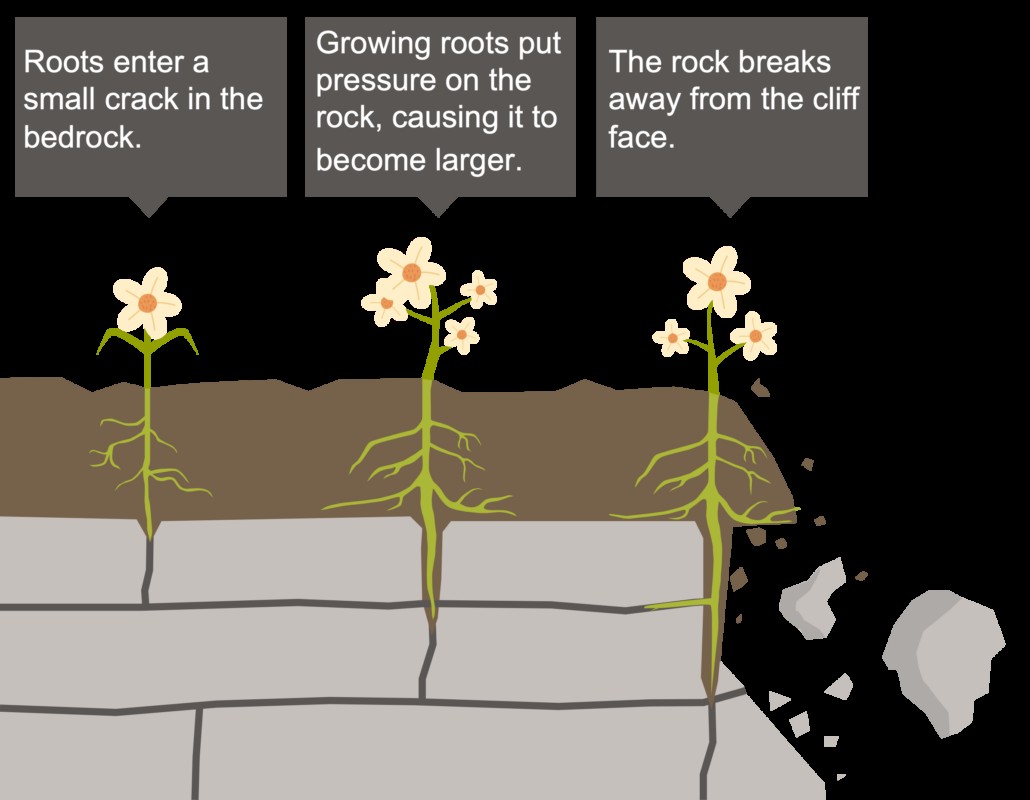 Biological Weathering Labelled
Biological Weathering Labelled
Alt Text: Illustration of biological weathering, showcasing how plant roots penetrate and break apart rocks, and how organisms contribute to rock decomposition.
3. Factors Affecting Chemical Weathering
The rate and extent of chemical weathering are influenced by several factors, including climate, rock composition, surface area, and the presence of vegetation.
3.1. Climate
Climate is one of the most critical factors affecting chemical weathering. Temperature and moisture levels can significantly influence the rate of chemical reactions.
-
Temperature: Higher temperatures generally accelerate chemical reaction rates, leading to faster weathering. However, some reactions may be inhibited at very high temperatures due to the evaporation of water.
-
Moisture: Water is essential for most chemical weathering processes. It acts as a solvent, transports reactants and products, and participates in hydrolysis and carbonation reactions. Regions with high rainfall and humidity tend to experience more intense chemical weathering.
3.2. Rock Composition
The mineral composition of a rock can influence its susceptibility to chemical weathering. Some minerals are more resistant to weathering than others, depending on their chemical structure and bonding.
-
Silicate Minerals: Silicate minerals, such as quartz and feldspar, vary in their resistance to weathering. Quartz is highly resistant, while feldspar is more susceptible to hydrolysis.
-
Carbonate Minerals: Carbonate minerals, such as calcite and dolomite, are readily dissolved by acidic solutions, making them vulnerable to carbonation.
-
Iron-Rich Minerals: Iron-rich minerals, such as pyrite and olivine, are easily oxidized, leading to the formation of rust and the breakdown of the rock.
3.3. Surface Area
The surface area of a rock exposed to weathering agents can significantly affect the rate of chemical weathering. The greater the surface area, the more contact the rock has with water, oxygen, and other reactive chemicals.
-
Fractures and Joints: Rocks with numerous fractures and joints have a larger surface area, allowing weathering agents to penetrate deeper into the rock and accelerate its breakdown.
-
Particle Size: Smaller rock particles have a larger surface area per unit volume than larger particles. This means that finely ground rocks weather more quickly than massive rocks.
3.4. Vegetation
Vegetation can influence chemical weathering in several ways. Plant roots can exert physical pressure on rocks, causing them to crack and break apart. Additionally, plants can release organic acids that dissolve minerals and promote chemical weathering.
-
Organic Acids: Plant roots and decaying organic matter release organic acids, such as citric acid and oxalic acid, which can dissolve minerals and enhance chemical weathering.
-
Soil Microbes: Soil microbes play a crucial role in the decomposition of organic matter and the release of nutrients. They also contribute to chemical weathering by producing organic acids and enzymes that dissolve minerals.
4. Examples of Chemical Weathering in Action
Chemical weathering is evident in various landscapes around the world, shaping distinctive landforms and influencing ecosystem processes.
4.1. Karst Landscapes
Karst landscapes are characterized by caves, sinkholes, and underground drainage systems, formed by the dissolution of limestone and other carbonate rocks. These landscapes are common in regions with abundant rainfall and soluble bedrock.
-
Caves: Caves are formed by the dissolution of limestone by acidic groundwater. Over time, the groundwater dissolves the rock, creating underground chambers and passageways.
-
Sinkholes: Sinkholes are depressions in the ground surface formed by the collapse of underlying caves or the dissolution of bedrock. They can range in size from a few meters to hundreds of meters in diameter.
-
Underground Rivers: Underground rivers are formed when surface water drains into sinkholes and flows through underground cave systems. These rivers can transport large amounts of water and sediment, contributing to the erosion and shaping of karst landscapes.
4.2. Weathering of Monuments and Buildings
Chemical weathering can cause significant damage to monuments and buildings made of stone, particularly those made of limestone, marble, and sandstone. Acid rain, air pollution, and biological activity can accelerate the weathering process, leading to the deterioration of historical and cultural landmarks.
-
Acid Rain: Acid rain is formed when sulfur dioxide ($SO_2$) and nitrogen oxides ($NO_x$) from industrial emissions react with water in the atmosphere, producing sulfuric acid and nitric acid. These acids can dissolve stone, causing it to crumble and erode.
-
Air Pollution: Air pollution can also contribute to the weathering of stone by depositing particulate matter on the surface, which can trap moisture and promote chemical reactions.
-
Biological Activity: Biological activity, such as the growth of lichens and mosses, can also damage stone by releasing organic acids that dissolve minerals and weaken the structure.
4.3. Soil Formation
Chemical weathering is a critical process in soil formation. It breaks down rocks and minerals, releasing essential nutrients and forming clay minerals that contribute to soil fertility and water retention.
-
Nutrient Release: Chemical weathering releases essential nutrients, such as potassium, calcium, and phosphorus, from rocks and minerals. These nutrients are vital for plant growth and support terrestrial ecosystems.
-
Clay Formation: Chemical weathering leads to the formation of clay minerals, which play a crucial role in soil fertility, water retention, and contaminant transport. Clay minerals have a high surface area and can adsorb nutrients and water, making them available to plants.
-
Soil Structure: Chemical weathering can also influence soil structure by promoting the aggregation of soil particles. The formation of clay minerals and the release of organic matter can bind soil particles together, creating a stable soil structure that resists erosion.
5. The Importance of Chemical Weathering
Chemical weathering is a fundamental process that shapes the Earth’s surface, influences geochemical cycles, and supports life.
5.1. Shaping Landscapes
Chemical weathering contributes to the formation of various landforms, including karst landscapes, canyons, and valleys. The selective weathering of different rock types can create distinctive topographic features and patterns.
5.2. Influencing Geochemical Cycles
Chemical weathering plays a vital role in regulating the composition of the atmosphere, oceans, and soils. It controls the release and uptake of various elements, influencing global climate and ecosystem processes.
5.3. Supporting Life
Chemical weathering provides essential nutrients for plant growth and supports terrestrial ecosystems. The breakdown of minerals releases elements such as potassium, calcium, and phosphorus, which are vital for plant nutrition.
6. Chemical Weathering vs. Physical Weathering
Weathering is the breaking down or dissolving of rocks and minerals on the surface of the Earth. It can be broadly classified into two main types: chemical weathering and physical weathering. While both processes contribute to the degradation of rocks, they operate through different mechanisms and produce distinct effects.
6.1. Key Differences
| Feature | Chemical Weathering | Physical Weathering |
|---|---|---|
| Definition | Decomposition of rocks through chemical reactions | Disintegration of rocks through physical processes |
| Mechanism | Alters the chemical composition of rocks | Breaks rocks into smaller pieces without changing composition |
| Agents | Water, acids, oxygen, biological activity | Temperature changes, frost action, abrasion |
| Effects | Formation of new minerals, dissolution of rocks | Increased surface area, cracks and fractures |
| Examples | Dissolution, hydrolysis, oxidation, carbonation | Freeze-thaw, exfoliation, abrasion |
| Dominant Climate | Warm, humid climates | Cold, dry climates |
6.2. Complementary Processes
Chemical and physical weathering often work together to break down rocks. Physical weathering can increase the surface area of rocks, making them more susceptible to chemical weathering. Chemical weathering can weaken rocks, making them more vulnerable to physical weathering.
For example, freeze-thaw weathering can create cracks in rocks, allowing water and acids to penetrate deeper and accelerate chemical weathering. Similarly, chemical weathering can dissolve minerals that bind rock particles together, making the rock more susceptible to abrasion and other physical weathering processes.
7. Addressing Common Misconceptions
Several common misconceptions surround chemical weathering, which can lead to misunderstandings about its nature and importance.
7.1. Chemical Weathering is Always Harmful
While chemical weathering can damage monuments and buildings, it is not always harmful. In many cases, it is a beneficial process that contributes to soil formation, nutrient release, and the regulation of geochemical cycles.
7.2. Chemical Weathering Only Occurs in Wet Climates
While moisture is essential for most chemical weathering processes, it can also occur in dry climates, albeit at a slower rate. For example, oxidation can occur in arid regions, leading to the formation of desert varnish on rock surfaces.
7.3. Chemical Weathering is a Fast Process
Chemical weathering is generally a slow process that occurs over long periods. However, the rate of chemical weathering can vary depending on factors such as climate, rock composition, and surface area.
8. Frequently Asked Questions (FAQs)
| Question | Answer |
|---|---|
| What are the main types of chemical weathering? | The main types include dissolution, hydrolysis, oxidation, carbonation, and biological weathering. Each involves different chemical reactions that break down rocks and minerals. |
| How does climate affect chemical weathering? | Climate significantly influences chemical weathering. Higher temperatures accelerate reactions, and moisture is essential for processes like hydrolysis and carbonation. Warm, humid climates typically see more intense chemical weathering. |
| Why is chemical weathering important? | It shapes the Earth’s surface, influences geochemical cycles, releases essential nutrients for plant growth, and contributes to soil formation. |
| What is the difference between chemical and physical weathering? | Chemical weathering alters the chemical composition of rocks, while physical weathering breaks rocks into smaller pieces without changing their composition. |
| How does acid rain contribute to chemical weathering? | Acid rain, containing sulfuric and nitric acids, dissolves stone and accelerates the weathering of monuments, buildings, and natural rock formations. |
| What role do plants play in chemical weathering? | Plants release organic acids that dissolve minerals and promote chemical weathering. Plant roots can also exert physical pressure, contributing to the breakdown of rocks. |
| Can chemical weathering occur in dry climates? | Yes, though at a slower rate. Oxidation, for example, can still occur, leading to the formation of desert varnish on rocks. |
| How does surface area affect chemical weathering? | The greater the surface area, the more contact the rock has with weathering agents. Rocks with fractures and smaller particles weather more quickly due to increased exposure. |
| What are karst landscapes, and how are they formed? | Karst landscapes are characterized by caves, sinkholes, and underground drainage systems. They are formed by the dissolution of limestone and other carbonate rocks by acidic groundwater. |
| What types of rocks are most susceptible to chemical weathering? | Carbonate rocks like limestone and chalk, and iron-rich minerals like pyrite and olivine, are particularly susceptible to chemical weathering due to their chemical properties and reactivity. |
9. Conclusion: The Dynamic Nature of Chemical Weathering
Chemical weathering is a dynamic and essential process that shapes the Earth’s surface, influences geochemical cycles, and supports life. By understanding the mechanisms and factors that control chemical weathering, we can better appreciate its role in the Earth system and mitigate its potential impacts on human structures and ecosystems.
Do you still have questions about chemical weathering or any other science topic? Don’t hesitate to ask! At WHAT.EDU.VN, we are dedicated to providing clear, accurate, and accessible answers to all your questions. Our community of experts is ready to help you explore the wonders of science and expand your knowledge.
Have a question? Ask it on WHAT.EDU.VN and get free answers today Address: 888 Question City Plaza, Seattle, WA 98101, United States. Whatsapp: +1 (206) 555-7890. Trang web: what.edu.vn