Chemiosmosis is the movement of ions across a selectively permeable membrane, down their electrochemical gradient. This process is essential for energy production in cells, and at WHAT.EDU.VN, we aim to provide you with a clear and easy-to-understand explanation of how it works. Learn about the chemiosmotic gradient, ATP synthesis, and mitochondrial function.
1. Understanding Chemiosmosis: The Basics
Chemiosmosis is a vital process in cellular respiration and photosynthesis, responsible for producing the majority of ATP, the energy currency of the cell. It involves the movement of ions, typically protons (H+), across a membrane, driven by an electrochemical gradient.
1.1. Defining Chemiosmosis
Chemiosmosis, derived from the Greek words “chemi” (chemical) and “osmos” (push), refers to the diffusion of ions across a selectively permeable membrane. This diffusion is coupled with a chemical reaction, typically the synthesis of ATP. The process relies on the electrochemical gradient, a combination of the concentration gradient and the electrical potential difference across the membrane.
1.2. The Electrochemical Gradient
The electrochemical gradient is a crucial aspect of chemiosmosis. It is formed by the uneven distribution of ions across the membrane, resulting in both a concentration gradient and an electrical gradient. The concentration gradient arises from the difference in ion concentration on either side of the membrane, while the electrical gradient is due to the difference in charge.
1.3. Key Players in Chemiosmosis
Several key components are involved in chemiosmosis:
- Electron Transport Chain (ETC): A series of protein complexes embedded in the membrane that facilitate the transfer of electrons, releasing energy in the process.
- Proton Pump: A protein that actively transports protons across the membrane, against their concentration gradient, using energy from the ETC.
- ATP Synthase: An enzyme that utilizes the electrochemical gradient to synthesize ATP from ADP and inorganic phosphate.
- Membrane: A selectively permeable barrier that separates the regions of different ion concentrations.
2. The Process of Chemiosmosis: A Step-by-Step Explanation
Chemiosmosis involves several coordinated steps that lead to the generation of ATP:
2.1. Electron Transport and Proton Pumping
The process begins with the electron transport chain (ETC). Electrons from molecules like NADH and FADH2 are passed along the chain, releasing energy. This energy is used to pump protons (H+) from one side of the membrane to the other, creating a high concentration of protons on one side and a low concentration on the other. This creates the electrochemical gradient.
2.2. Formation of the Proton Gradient
As protons are pumped across the membrane, they accumulate, forming a high concentration on one side. This creates a proton gradient, also known as a pH gradient or an electrochemical gradient. This gradient stores potential energy, similar to water held behind a dam.
2.3. ATP Synthesis by ATP Synthase
The potential energy stored in the proton gradient is then harnessed by ATP synthase, a remarkable enzyme that acts as a molecular motor. Protons flow down their electrochemical gradient, passing through ATP synthase. This flow causes ATP synthase to rotate, converting the energy of the proton gradient into mechanical energy, which is then used to bind ADP and inorganic phosphate together to form ATP.
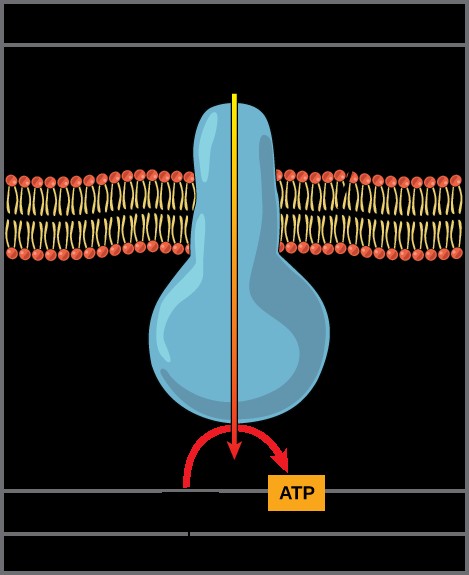 ATP Synthase: A complex, molecular machine that uses a proton (H+) gradient to form ATP from ADP and inorganic phosphate (Pi)
ATP Synthase: A complex, molecular machine that uses a proton (H+) gradient to form ATP from ADP and inorganic phosphate (Pi)
3. Chemiosmosis in Cellular Respiration
Cellular respiration is the process by which cells break down glucose to generate energy. Chemiosmosis plays a critical role in the final stage of cellular respiration, oxidative phosphorylation.
3.1. Oxidative Phosphorylation
Oxidative phosphorylation is the process where ATP is synthesized using the energy released during the electron transport chain. It occurs in the inner mitochondrial membrane of eukaryotic cells.
3.2. The Role of the Electron Transport Chain
The electron transport chain (ETC) in the inner mitochondrial membrane is crucial for chemiosmosis. Electrons from NADH and FADH2 are passed along the chain, releasing energy. This energy is used to pump protons from the mitochondrial matrix to the intermembrane space, creating the electrochemical gradient.
3.3. ATP Production in Mitochondria
The electrochemical gradient created by the ETC is then used by ATP synthase to produce ATP. As protons flow back into the mitochondrial matrix through ATP synthase, ATP is synthesized. This process generates the majority of ATP during cellular respiration.
4. Chemiosmosis in Photosynthesis
Photosynthesis is the process by which plants and other organisms convert light energy into chemical energy. Chemiosmosis is also essential in the light-dependent reactions of photosynthesis.
4.1. The Light-Dependent Reactions
The light-dependent reactions occur in the thylakoid membranes of chloroplasts. Light energy is absorbed by chlorophyll, which energizes electrons. These electrons are passed along an electron transport chain, releasing energy.
4.2. Proton Gradient Formation in Chloroplasts
As electrons move along the electron transport chain in the thylakoid membrane, protons are pumped from the stroma into the thylakoid lumen, creating a proton gradient.
4.3. ATP Synthesis in Chloroplasts
The proton gradient in the thylakoid lumen is then used by ATP synthase to produce ATP. As protons flow back into the stroma through ATP synthase, ATP is synthesized. This ATP is used in the light-independent reactions (Calvin cycle) to convert carbon dioxide into glucose.
5. Comparing Chemiosmosis in Mitochondria and Chloroplasts
While chemiosmosis occurs in both mitochondria and chloroplasts, there are some key differences:
| Feature | Mitochondria | Chloroplasts |
|---|---|---|
| Location | Inner mitochondrial membrane | Thylakoid membrane |
| Energy Source | Electrons from NADH and FADH2 | Light energy |
| Proton Pumping | From matrix to intermembrane space | From stroma to thylakoid lumen |
| ATP Use | Cellular respiration | Calvin cycle (light-independent reactions) |
| Primary Function | ATP production for cellular energy | ATP production for sugar synthesis |
6. The Significance of Chemiosmosis in Energy Production
Chemiosmosis is a highly efficient mechanism for energy production in cells. It allows cells to convert the energy stored in electrochemical gradients into the readily usable form of ATP.
6.1. Efficiency of ATP Production
Chemiosmosis is responsible for generating the vast majority of ATP in aerobic organisms. In cellular respiration, it produces significantly more ATP than glycolysis or the citric acid cycle alone.
6.2. Importance in Various Organisms
Chemiosmosis is essential for the survival of a wide range of organisms, including bacteria, archaea, plants, and animals. It is a fundamental process that underpins life as we know it.
6.3. Evolutionary Perspective
The evolution of chemiosmosis was a pivotal event in the history of life. It allowed organisms to harness energy more efficiently, leading to the development of more complex life forms.
7. Factors Affecting Chemiosmosis
Several factors can influence the efficiency of chemiosmosis:
7.1. Membrane Integrity
The integrity of the membrane is crucial for maintaining the electrochemical gradient. If the membrane is damaged or leaky, protons may diffuse across it, reducing the gradient and decreasing ATP production.
7.2. Proton Gradient Strength
The strength of the proton gradient directly affects the rate of ATP synthesis. Factors that influence the proton pumping rate or the rate of proton leakage can affect the gradient strength.
7.3. Availability of Electron Carriers
The availability of electron carriers like NADH and FADH2 can limit the rate of electron transport and proton pumping.
7.4. Inhibitors and Uncouplers
Certain substances can inhibit or uncouple chemiosmosis. Inhibitors block the electron transport chain, preventing proton pumping, while uncouplers allow protons to leak across the membrane, dissipating the gradient.
8. Chemiosmosis and Human Health
Chemiosmosis plays a vital role in human health, as it is essential for energy production in cells. Disruptions in chemiosmosis can lead to various health problems.
8.1. Mitochondrial Diseases
Mitochondrial diseases are a group of disorders caused by defects in mitochondrial function. These defects can impair chemiosmosis, leading to reduced ATP production and a variety of symptoms, including muscle weakness, fatigue, and neurological problems.
8.2. Aging and Chemiosmosis
As we age, mitochondrial function tends to decline, which can affect chemiosmosis. This decline in energy production may contribute to age-related diseases and overall aging.
8.3. Therapeutic Interventions
Researchers are exploring various therapeutic interventions to improve mitochondrial function and enhance chemiosmosis. These interventions include dietary modifications, exercise, and drugs that target mitochondrial processes.
9. Chemiosmosis Research and Future Directions
Chemiosmosis remains an active area of research, with scientists exploring its intricacies and potential applications.
9.1. Current Research Trends
Current research trends include investigating the structure and function of ATP synthase, exploring the mechanisms of proton pumping, and developing new therapies for mitochondrial diseases.
9.2. Potential Applications
Potential applications of chemiosmosis research include developing new energy technologies, improving crop yields, and treating human diseases.
9.3. Future Directions
Future directions in chemiosmosis research may focus on understanding the regulation of chemiosmosis, exploring its role in various cellular processes, and developing new tools to study it.
10. Common Misconceptions About Chemiosmosis
Several misconceptions exist regarding chemiosmosis:
10.1. Chemiosmosis Only Occurs in Mitochondria
While chemiosmosis is well-known for its role in mitochondria, it also occurs in chloroplasts and bacteria.
10.2. Chemiosmosis is the Same as Osmosis
Chemiosmosis involves the movement of ions across a membrane, coupled with a chemical reaction, while osmosis involves the movement of water across a membrane, driven by differences in water potential.
10.3. ATP Synthase is a Simple Channel
ATP synthase is a complex molecular machine that uses the flow of protons to generate ATP. It is not simply a channel that allows protons to pass through.
11. Chemiosmosis: A Summary
Chemiosmosis is the movement of ions across a selectively permeable membrane, down their electrochemical gradient. It is essential for ATP production in cellular respiration and photosynthesis. The process involves the electron transport chain, proton pumping, and ATP synthase. Chemiosmosis is influenced by membrane integrity, proton gradient strength, and the availability of electron carriers. Disruptions in chemiosmosis can lead to various health problems.
12. FAQ About Chemiosmosis
| Question | Answer |
|---|---|
| What exactly is chemiosmosis? | Chemiosmosis is the movement of ions across a selectively permeable membrane, down their electrochemical gradient, coupled with ATP synthesis. |
| Where does chemiosmosis occur? | Chemiosmosis occurs in the inner mitochondrial membrane (cellular respiration) and the thylakoid membrane of chloroplasts (photosynthesis). |
| What is the role of ATP synthase? | ATP synthase is an enzyme that uses the electrochemical gradient to synthesize ATP from ADP and inorganic phosphate. |
| What is the electron transport chain? | The electron transport chain is a series of protein complexes that transfer electrons, releasing energy used to pump protons across the membrane. |
| How is the proton gradient formed? | The proton gradient is formed by pumping protons across the membrane, creating a high concentration of protons on one side and a low concentration on the other. |
| What is oxidative phosphorylation? | Oxidative phosphorylation is the process where ATP is synthesized using the energy released during the electron transport chain in mitochondria. |
| How does chemiosmosis relate to energy? | Chemiosmosis allows cells to convert the energy stored in electrochemical gradients into the readily usable form of ATP, which powers various cellular processes. |
| What factors affect chemiosmosis? | Factors include membrane integrity, proton gradient strength, availability of electron carriers, and the presence of inhibitors or uncouplers. |
| Can chemiosmosis be disrupted? | Yes, disruptions in chemiosmosis can lead to various health problems, such as mitochondrial diseases. |
| What is the significance of chemiosmosis? | Chemiosmosis is essential for the survival of a wide range of organisms, as it is a fundamental process that underpins life by efficiently producing ATP. |
13. More Questions About Chemiosmosis Answered
Let’s dive deeper into some frequently asked questions about chemiosmosis to further clarify its significance and function.
13.1. How does the electron transport chain contribute to chemiosmosis?
The electron transport chain (ETC) is pivotal in chemiosmosis. As electrons move through the chain, energy is released. This energy is then used to pump protons (H+) across the inner mitochondrial membrane (in cellular respiration) or the thylakoid membrane (in photosynthesis), creating a proton gradient. The ETC, therefore, establishes the electrochemical gradient necessary for ATP synthesis.
13.2. What happens if ATP synthase is inhibited?
If ATP synthase is inhibited, the flow of protons down the electrochemical gradient is blocked. This prevents the synthesis of ATP, which is crucial for cellular energy. Consequently, cells would need to rely on less efficient energy-producing processes like glycolysis, leading to a significant reduction in ATP production. This can result in cellular dysfunction and, in severe cases, cell death.
13.3. Can chemiosmosis occur without a membrane?
No, chemiosmosis cannot occur without a membrane. The selectively permeable membrane is essential for creating and maintaining the electrochemical gradient. It separates regions of different ion concentrations, allowing for the build-up of protons on one side and their subsequent flow through ATP synthase on the other side.
13.4. What are some practical applications of understanding chemiosmosis?
Understanding chemiosmosis has several practical applications. For instance, it can aid in the development of therapies for mitochondrial diseases by targeting specific defects in the electron transport chain or ATP synthase. It can also inform strategies to improve crop yields by optimizing photosynthetic efficiency. Additionally, understanding chemiosmosis can contribute to the development of new energy technologies, such as bio-batteries that mimic the process of ATP synthesis.
13.5. How does chemiosmosis relate to the concept of free energy?
Chemiosmosis is closely related to the concept of free energy. The electron transport chain releases free energy as electrons move through it. This free energy is then used to pump protons against their concentration gradient, storing potential energy in the form of an electrochemical gradient. ATP synthase then converts this potential energy back into a usable form of free energy, ATP, which can be used to drive cellular processes.
13.6. What is the role of NADH and FADH2 in chemiosmosis?
NADH and FADH2 are electron carriers that donate electrons to the electron transport chain. They are produced during glycolysis and the citric acid cycle. These electrons provide the energy needed to pump protons across the membrane, establishing the electrochemical gradient necessary for ATP synthesis. Without NADH and FADH2, the electron transport chain would not function, and chemiosmosis would not occur.
13.7. How does the pH gradient contribute to chemiosmosis?
The pH gradient is a component of the electrochemical gradient. As protons are pumped across the membrane, the pH decreases on one side (higher concentration of protons) and increases on the other side (lower concentration of protons). This pH gradient contributes to the overall electrochemical gradient, which drives the flow of protons through ATP synthase.
13.8. What are some examples of chemiosmosis inhibitors and uncouplers?
Inhibitors of chemiosmosis block the electron transport chain, preventing proton pumping. Examples include cyanide and rotenone. Uncouplers, on the other hand, allow protons to leak across the membrane, dissipating the proton gradient. An example is dinitrophenol (DNP). Both inhibitors and uncouplers reduce ATP production.
13.9. How does chemiosmosis differ between aerobic and anaerobic organisms?
In aerobic organisms, chemiosmosis is coupled with the electron transport chain that uses oxygen as the final electron acceptor. In anaerobic organisms, chemiosmosis can occur with different electron transport chains that use other electron acceptors, such as sulfate or nitrate. The specific components of the electron transport chain and the electron acceptors vary depending on the organism and its environment.
13.10. What are the key differences between chemiosmosis in photosynthesis and cellular respiration?
| Feature | Photosynthesis | Cellular Respiration |
|---|---|---|
| Location | Thylakoid membrane of chloroplasts | Inner mitochondrial membrane |
| Energy Source | Light energy | Chemical energy from NADH and FADH2 |
| Proton Pumping Direction | From stroma to thylakoid lumen | From mitochondrial matrix to intermembrane space |
| Final Electron Acceptor | NADP+ | Oxygen |
| Purpose | ATP and NADPH production for carbon fixation (Calvin cycle) | ATP production for cellular energy |
| Key Molecules | Water, carbon dioxide, chlorophyll | Glucose, oxygen |
| Organisms | Plants, algae, cyanobacteria | Animals, plants, fungi, bacteria |
14. Expert Insights on Chemiosmosis
To provide a deeper understanding of chemiosmosis, let’s explore insights from leading experts in the field.
14.1. Dr. Peter Mitchell’s Chemiosmotic Theory
Dr. Peter Mitchell, who was awarded the Nobel Prize in Chemistry in 1978, proposed the chemiosmotic theory. This theory revolutionized our understanding of how ATP is produced in cells. Mitchell’s theory explained that the energy for ATP synthesis comes from an electrochemical gradient of protons across a membrane, rather than direct chemical coupling. This groundbreaking idea was initially met with skepticism but eventually became widely accepted.
14.2. Current Research by Leading Scientists
Current research on chemiosmosis is being conducted by scientists around the world. They are investigating the structure and function of ATP synthase, exploring the mechanisms of proton pumping, and developing new therapies for mitochondrial diseases. Some researchers are also exploring the role of chemiosmosis in various cellular processes, such as signal transduction and cell differentiation.
14.3. Emerging Trends in Chemiosmosis Research
Emerging trends in chemiosmosis research include the use of advanced imaging techniques to visualize the process in real-time, the development of new computational models to simulate the behavior of ATP synthase, and the exploration of the potential of chemiosmosis for bioenergy applications. These advances are helping us to gain a more complete understanding of this fundamental process.
15. Optimizing Your Understanding of Chemiosmosis
Understanding chemiosmosis can be complex, but there are several strategies you can use to enhance your comprehension.
15.1. Visual Aids and Diagrams
Visual aids, such as diagrams and animations, can be very helpful for understanding chemiosmosis. These visuals can illustrate the movement of electrons, protons, and ATP synthase in a way that is easy to grasp.
15.2. Interactive Simulations
Interactive simulations allow you to explore the process of chemiosmosis in a hands-on way. You can adjust various parameters, such as the proton gradient or the concentration of electron carriers, and see how these changes affect ATP production.
15.3. Real-World Examples
Connecting chemiosmosis to real-world examples can make the concept more relatable. For example, you can discuss how chemiosmosis is essential for muscle function or how disruptions in chemiosmosis can lead to mitochondrial diseases.
15.4. Further Reading and Resources
There are many excellent resources available for learning more about chemiosmosis, including textbooks, scientific articles, and online tutorials. Exploring these resources can help you to deepen your understanding of the topic.
16. Let WHAT.EDU.VN Answer All Your Questions
We hope this comprehensive guide has clarified your understanding of chemiosmosis. However, if you still have questions, don’t hesitate to ask! At WHAT.EDU.VN, we provide a platform where you can ask any question and receive free answers from knowledgeable experts.
16.1. Why Choose WHAT.EDU.VN?
WHAT.EDU.VN offers a unique service that connects you with a community of experts ready to answer your questions. Our platform is easy to use, completely free, and dedicated to providing accurate and reliable information.
16.2. How to Ask a Question on WHAT.EDU.VN
Asking a question on WHAT.EDU.VN is simple. Just visit our website, type your question into the search bar, and submit. Our community of experts will review your question and provide you with a detailed answer.
16.3. Benefits of Using WHAT.EDU.VN
- Free Answers: Get your questions answered without any cost.
- Expert Knowledge: Connect with knowledgeable experts in various fields.
- Quick Responses: Receive timely answers to your questions.
- Easy to Use: Our platform is user-friendly and straightforward.
17. Contact Us
If you have any questions or need further assistance, please feel free to contact us:
- Address: 888 Question City Plaza, Seattle, WA 98101, United States
- WhatsApp: +1 (206) 555-7890
- Website: WHAT.EDU.VN
18. Call to Action
Do you have more questions about chemiosmosis or any other topic? Visit WHAT.EDU.VN today and ask away! Our community of experts is ready to provide you with free and accurate answers. Don’t struggle with unanswered questions – let what.edu.vn help you find the information you need.

