Distillation is a process that involves vaporizing a liquid and then condensing the vapor back into liquid form to separate components based on their boiling points. At WHAT.EDU.VN, we offer clear explanations and guidance to help you understand complex topics like distillation. This process is crucial in various industries, from creating alcoholic beverages to purifying chemicals. Explore the science behind separation techniques with us!
1. Defining Distillation: The Basics
Distillation is a separation technique used to separate mixtures of liquids with different boiling points. The process involves heating the mixture to a temperature that vaporizes the most volatile component. The vapor is then cooled and condensed back into a liquid, which is collected separately. This process can be repeated to achieve higher purity. This efficient separation is used across many industries.
1.1. What is the Fundamental Principle Behind Distillation?
The fundamental principle behind distillation lies in the differences in boiling points of the substances within a mixture. When a mixture is heated, the substance with the lower boiling point will vaporize first. The vapor is then cooled, causing it to condense back into a liquid, which can be collected separately. This allows for the separation of components based on their volatility.
1.2. What are the Key Components of a Distillation Setup?
A basic distillation setup includes several key components:
- Still or Retort: The container where the liquid mixture is heated.
- Heat Source: A device to provide the necessary heat for vaporization (e.g., a Bunsen burner or heating mantle).
- Condenser: A device to cool the vapor and convert it back into a liquid.
- Receiver: A container to collect the condensed liquid, known as the distillate.
- Thermometer: Used to monitor the temperature of the vapor, ensuring accurate separation.
1.3. What Types of Mixtures are Suitable for Distillation?
Distillation is best suited for separating mixtures of liquids with significantly different boiling points. It can also be used to separate volatile liquids from non-volatile solids. Common examples include:
- Separating ethanol from water in the production of alcoholic beverages.
- Purifying solvents in chemical laboratories.
- Desalination of seawater to produce fresh water.
- Fractional distillation of crude oil to separate gasoline, kerosene, and other petroleum products.
2. Types of Distillation Techniques
Several types of distillation techniques exist, each designed for specific applications and mixture types. Understanding these variations can help in selecting the most appropriate method for a given separation task.
2.1. What is Simple Distillation and When is it Used?
Simple distillation is the most basic form of distillation, suitable for separating liquids with large differences in boiling points (typically greater than 25°C). The process involves heating the mixture, collecting the vapor that rises, and then condensing it back into a liquid in a separate container. It is commonly used in laboratory settings for purifying solvents or separating a volatile liquid from a non-volatile solute.
2.2. What is Fractional Distillation and How Does It Work?
Fractional distillation is used to separate liquids with closer boiling points by using a fractionating column. This column is packed with glass beads or metal pieces, providing a large surface area for repeated vaporization and condensation cycles. The component with the lower boiling point will gradually move to the top of the column, while the higher boiling point component condenses and flows back down. This continuous process allows for a more efficient separation of the liquids.
2.3. What is Steam Distillation and its Applications?
Steam distillation is used to separate temperature-sensitive compounds that might decompose at high temperatures. In this method, steam is passed through the mixture, causing the volatile components to vaporize at a lower temperature than their boiling points. The vapor mixture is then cooled and condensed, and the components are separated. This technique is particularly useful for extracting essential oils from plants.
2.4. What is Vacuum Distillation and Why is it Necessary?
Vacuum distillation is used for liquids that have very high boiling points or that decompose when heated to their boiling points under atmospheric pressure. By reducing the pressure, the boiling point of the liquid is lowered, allowing it to vaporize at a lower temperature. This prevents decomposition and makes it possible to distill high-boiling substances.
2.5. What is Multiple-Effect Distillation and Where is it Employed?
Multiple-effect distillation, also known as multistage flash evaporation, is primarily used in large commercial desalination plants. This process involves passing a liquid from a container under high pressure to one under lower pressure, causing rapid vaporization. The resulting vapor is then condensed into distillate. This method is energy-efficient as the heat released during condensation in one stage is used for vaporization in another stage.
3. Industrial Applications of Distillation
Distillation plays a crucial role in various industries, enabling the production of essential products and the purification of valuable substances. Its versatility and efficiency make it an indispensable technique in many industrial processes.
3.1. How is Distillation Used in Petroleum Refining?
In petroleum refining, fractional distillation is used to separate crude oil into various components such as gasoline, kerosene, diesel, and lubricating oils. Crude oil is heated, and the vapors are passed through a fractionating column. Each component condenses at different levels within the column based on its boiling point, allowing for the efficient separation of valuable petroleum products.
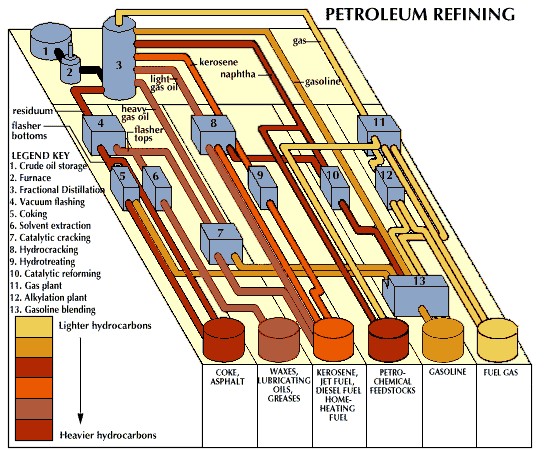 Fractional distillation of crude oil separates components
Fractional distillation of crude oil separates components
3.2. How is Distillation Applied in the Production of Alcoholic Beverages?
Distillation is a key step in the production of many alcoholic beverages, such as whiskey, vodka, and gin. After fermentation, the resulting mixture contains ethanol and water, along with other compounds. Distillation is used to increase the alcohol content by separating the ethanol from the water and other impurities, resulting in a higher-proof spirit.
3.3. What is the Role of Distillation in Chemical Industries?
In the chemical industry, distillation is used for purifying solvents, separating reaction products, and recovering valuable chemicals. It ensures the purity of reagents and products, which is essential for various chemical processes. Distillation helps in achieving high-quality standards and efficient production in the chemical industry.
3.4. How Does Distillation Contribute to Water Purification and Desalination?
Distillation is an effective method for water purification, especially in desalination plants. Seawater is heated, and the resulting steam is collected and condensed to produce fresh water. This process removes salts and other impurities, providing a reliable source of potable water in regions where freshwater resources are scarce.
3.5. What are Some Other Notable Industrial Applications of Distillation?
Besides the major applications, distillation is also used in:
- Pharmaceutical Industry: Purification of drugs and pharmaceutical intermediates.
- Essential Oil Extraction: Extraction of essential oils from plants for use in perfumes, flavors, and aromatherapy.
- Food and Beverage Industry: Concentration of fruit juices and production of flavor extracts.
- Cosmetics Industry: Purification of ingredients used in cosmetic products.
4. The Science Behind Distillation: A Deeper Look
To fully appreciate the power of distillation, it is essential to understand the scientific principles that govern the process. This includes the concepts of vapor pressure, boiling point, and phase equilibrium.
4.1. What is Vapor Pressure and How Does it Affect Distillation?
Vapor pressure is the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases (solid or liquid) at a given temperature. In distillation, substances with higher vapor pressures at a given temperature will vaporize more readily. This difference in vapor pressure is what allows for the separation of components in a mixture.
4.2. How Does Boiling Point Relate to the Efficiency of Distillation?
The boiling point of a substance is the temperature at which its vapor pressure equals the surrounding atmospheric pressure. Substances with lower boiling points vaporize more easily, making them easier to separate through distillation. The larger the difference in boiling points between the components of a mixture, the more efficient the distillation process will be.
4.3. What is Azeotropic Distillation and When is it Needed?
Azeotropic distillation is a specialized technique used when the components of a mixture form an azeotrope, which is a mixture that has a constant boiling point and cannot be separated by simple or fractional distillation. In azeotropic distillation, a third component (an entrainer) is added to alter the vapor pressures of the original components, breaking the azeotrope and allowing for separation.
4.4. What Factors Affect the Purity of the Distillate?
Several factors can affect the purity of the distillate:
- Boiling Point Differences: Larger differences lead to higher purity.
- Distillation Rate: Slower rates allow for better separation.
- Column Efficiency: Fractionating columns with higher surface areas improve separation.
- Reflux Ratio: The ratio of condensed liquid returned to the column versus collected as distillate; higher ratios improve purity.
- Temperature Control: Precise temperature control ensures accurate separation of components.
4.5. How Does Pressure Influence the Distillation Process?
Pressure plays a significant role in distillation. Reducing the pressure lowers the boiling points of the liquids, making it possible to distill substances at lower temperatures, thus preventing decomposition. This is the principle behind vacuum distillation, which is used for heat-sensitive compounds.
5. Practical Distillation Techniques for Beginners
For those new to distillation, understanding the basic techniques and equipment is crucial. Here are some practical tips to get started with simple distillation.
5.1. What Equipment is Needed for a Basic Distillation Setup?
A basic distillation setup includes:
- Round-bottom flask: To hold the liquid mixture.
- Heating mantle or Bunsen burner: To provide heat.
- Distillation head: To connect the flask to the condenser.
- Thermometer adapter and thermometer: To monitor vapor temperature.
- Condenser: To cool and condense the vapor.
- Receiving flask: To collect the distillate.
- Clamps and stands: To support the apparatus.
5.2. How to Set Up a Simple Distillation Apparatus?
- Assemble the Apparatus: Securely clamp the round-bottom flask, distillation head, condenser, and receiving flask.
- Connect the Condenser: Attach water hoses to the condenser, ensuring water flows in from the bottom and out from the top.
- Add the Mixture: Pour the liquid mixture into the round-bottom flask.
- Insert the Thermometer: Place the thermometer in the distillation head, ensuring the bulb is positioned correctly to measure the vapor temperature.
- Apply Heat: Begin heating the flask gently, monitoring the temperature.
- Collect the Distillate: As the vapor reaches the condenser, it will condense and drip into the receiving flask. Collect the distillate in fractions based on temperature ranges.
5.3. What are Some Safety Precautions to Keep in Mind?
- Wear Safety Goggles: Protect your eyes from splashes or chemical vapors.
- Use a Fume Hood: Perform distillation in a well-ventilated area to avoid inhaling harmful vapors.
- Avoid Overheating: Heat the mixture gently to prevent bumping and potential hazards.
- Never Distill to Dryness: Stop heating when a small amount of liquid remains in the flask to prevent overheating and potential explosions.
- Proper Disposal: Dispose of chemical waste properly according to laboratory guidelines.
5.4. How to Troubleshoot Common Distillation Problems?
- No Distillate: Ensure the apparatus is properly sealed, the heat source is adequate, and the thermometer is correctly positioned.
- Low Purity: Increase the reflux ratio, slow down the distillation rate, or use a fractionating column.
- Bumping: Use boiling chips or stir the mixture to ensure smooth boiling.
- Condenser Not Cooling: Check the water flow and ensure the condenser is not clogged.
5.5. What are Some Tips for Achieving Better Separation?
- Slow Distillation Rate: A slower rate allows for better separation of components.
- Use a Fractionating Column: For mixtures with close boiling points, a fractionating column is essential.
- Maintain a Constant Temperature: Keep the temperature steady to ensure accurate separation.
- Collect Fractions: Collect the distillate in separate fractions based on temperature ranges to improve purity.
- Optimize Reflux Ratio: Adjust the reflux ratio to achieve the best balance between purity and yield.
6. Advanced Distillation Techniques and Innovations
Beyond the basic methods, several advanced distillation techniques and innovations have been developed to address specific challenges and improve efficiency.
6.1. What is Short Path Distillation and its Advantages?
Short path distillation is a technique used for compounds with very high boiling points or those that are heat-sensitive. The key feature is the short distance the vapor travels from the heating flask to the condenser, minimizing thermal degradation. This method is particularly useful in the pharmaceutical and cannabis industries for isolating and purifying specific compounds.
6.2. How Does Extractive Distillation Enhance Separation?
Extractive distillation is used when simple or azeotropic distillation is not sufficient to separate a mixture. A solvent is added to the mixture, which selectively interacts with one of the components, altering its volatility and making it easier to separate through distillation. This technique is often used in the petrochemical industry.
6.3. What is Membrane Distillation and its Applications?
Membrane distillation is a hybrid process that combines distillation with membrane separation. A hydrophobic membrane separates two liquid phases, and vapor passes through the membrane due to a vapor pressure difference. This technique is used in desalination, wastewater treatment, and the food industry.
6.4. How Does Reactive Distillation Combine Reaction and Separation?
Reactive distillation combines chemical reaction and separation in a single unit. This is particularly useful when the reaction is equilibrium-limited or when the product needs to be quickly removed to prevent side reactions. The process enhances reaction conversion and simplifies the overall process, making it more efficient and cost-effective.
6.5. What are Some Emerging Trends in Distillation Technology?
Emerging trends in distillation technology include:
- Energy Efficiency: Development of more energy-efficient distillation processes to reduce operating costs and environmental impact.
- Process Intensification: Combining multiple unit operations into a single unit to reduce equipment size and capital costs.
- Advanced Control Systems: Implementation of advanced control systems to optimize distillation processes and improve product quality.
- Sustainable Distillation: Use of renewable energy sources and waste heat recovery to make distillation more sustainable.
- Micro-Distillation: Development of micro-distillation systems for small-scale applications, such as in research laboratories and point-of-use applications.
7. Common Misconceptions About Distillation
It is essential to address some common misconceptions about distillation to ensure a clear understanding of the process.
7.1. Is Distillation Only Used for Alcohol Production?
While distillation is widely used in alcohol production, it has numerous other applications, including petroleum refining, chemical purification, water desalination, and essential oil extraction.
7.2. Does Distillation Always Result in Pure Substances?
Distillation can significantly improve the purity of substances, but achieving absolute purity is often challenging. Factors such as boiling point differences, distillation rate, and column efficiency can affect the final purity.
7.3. Is Distillation a Simple and Easy Process?
While the basic principles of distillation are straightforward, achieving efficient and high-purity separation can be complex. Advanced techniques and careful control of parameters are often necessary.
7.4. Can Any Liquid Mixture be Separated by Distillation?
Distillation is most effective for mixtures of liquids with significantly different boiling points. Azeotropic mixtures require specialized techniques like azeotropic distillation, and some mixtures may not be suitable for distillation at all.
7.5. Is Distillation Always Energy-Efficient?
Traditional distillation processes can be energy-intensive. However, modern techniques like multiple-effect distillation and heat integration strategies are designed to improve energy efficiency.
8. Distillation in Everyday Life
Distillation is not just a process confined to laboratories and industrial plants; it plays a role in many aspects of everyday life.
8.1. How is Distillation Used in Home Brewing and Winemaking?
Home brewers and winemakers use distillation to produce spirits and fortified wines. While regulations vary by location, the basic principles of distillation are applied to increase the alcohol content and refine the flavors of these beverages.
8.2. How Does Distillation Contribute to Food and Beverage Production?
Distillation is used in the food and beverage industry for various purposes, such as concentrating fruit juices, producing flavor extracts, and purifying water used in food processing.
8.3. What Role Does Distillation Play in the Production of Essential Oils?
Steam distillation is a common method for extracting essential oils from plants. These oils are used in perfumes, aromatherapy, and various cosmetic and household products.
8.4. How is Distilled Water Used in Households?
Distilled water is used in households for various purposes, such as:
- Steam Irons: To prevent mineral buildup.
- Humidifiers: To avoid dispersing minerals into the air.
- Automotive Applications: In car batteries and cooling systems.
- Medical Devices: In CPAP machines and nebulizers.
8.5. How Does Distillation Help in Waste Management and Recycling?
Distillation can be used in waste management and recycling to recover valuable solvents from industrial waste streams. This helps reduce waste and promotes sustainable practices.
9. Future Trends in Distillation Technology
The field of distillation is continuously evolving, with ongoing research and development aimed at improving efficiency, sustainability, and applicability.
9.1. What are the Latest Research and Development Efforts in Distillation?
Current research efforts are focused on:
- Novel Distillation Techniques: Exploring new distillation methods with improved performance and energy efficiency.
- Process Optimization: Developing advanced control systems and optimization algorithms to improve distillation processes.
- Sustainable Distillation: Investigating the use of renewable energy sources and waste heat recovery to make distillation more sustainable.
- Integration with Other Processes: Combining distillation with other separation and reaction processes to create more efficient and integrated systems.
- Micro- and Nano-Distillation: Developing micro- and nano-distillation systems for small-scale applications and high-throughput screening.
9.2. How is Artificial Intelligence (AI) Being Used in Distillation Processes?
AI is being used in distillation processes for:
- Process Monitoring and Control: Developing AI-based systems to monitor and control distillation processes in real-time, optimizing performance and preventing upsets.
- Process Modeling and Simulation: Using AI to develop accurate models and simulations of distillation processes, enabling better design and optimization.
- Fault Detection and Diagnosis: Implementing AI-based systems to detect and diagnose faults in distillation equipment, improving reliability and reducing downtime.
- Predictive Maintenance: Using AI to predict when maintenance is needed, preventing costly breakdowns and extending the lifespan of equipment.
- Process Optimization: Applying AI algorithms to optimize distillation processes, maximizing yield, minimizing energy consumption, and improving product quality.
9.3. What Role Will Nanotechnology Play in Future Distillation Technologies?
Nanotechnology is expected to play a significant role in future distillation technologies through:
- Enhanced Membranes: Developing nanomaterials for use in membrane distillation, improving membrane performance and selectivity.
- Catalytic Materials: Using nanomaterials as catalysts in reactive distillation, enhancing reaction rates and selectivity.
- Sensors and Monitoring: Developing nanosensors for real-time monitoring of distillation processes, enabling better control and optimization.
- Heat Transfer Enhancement: Using nanofluids to improve heat transfer in distillation equipment, increasing energy efficiency.
- Separation Materials: Developing novel nanomaterials for use in distillation columns, improving separation efficiency and reducing energy consumption.
9.4. How Will Distillation Contribute to a More Sustainable Future?
Distillation can contribute to a more sustainable future through:
- Energy Efficiency: Implementing energy-efficient distillation techniques and technologies to reduce energy consumption and greenhouse gas emissions.
- Waste Heat Recovery: Recovering and reusing waste heat from distillation processes to reduce energy consumption and improve overall efficiency.
- Use of Renewable Energy: Utilizing renewable energy sources, such as solar and geothermal energy, to power distillation processes.
- Waste Minimization: Developing distillation processes that minimize waste generation and promote the recovery and reuse of valuable materials.
- Water Purification: Using distillation for water purification and desalination, providing access to clean and safe drinking water.
9.5. What are the Potential Applications of Distillation in Space Exploration?
Distillation has potential applications in space exploration, including:
- Water Recycling: Recycling water from urine and other waste streams to provide potable water for astronauts.
- Life Support Systems: Producing oxygen and other essential gases for life support systems.
- Resource Utilization: Extracting and purifying valuable resources from lunar or Martian soil.
- Fuel Production: Producing rocket fuel from available resources on other planets.
- Waste Management: Processing and recycling waste materials to reduce the need for resupply missions.
10. Frequently Asked Questions (FAQs) About Distillation
Here are some frequently asked questions about distillation, covering various aspects of the process.
| Question | Answer |
|---|---|
| What is the primary purpose of distillation? | To separate components of a liquid mixture based on differences in boiling points. |
| How does simple distillation differ from fractional distillation? | Simple distillation is used for liquids with large boiling point differences, while fractional distillation is used for liquids with closer boiling points, employing a fractionating column for better separation. |
| What is steam distillation used for? | To separate temperature-sensitive compounds that might decompose at high temperatures, often used in extracting essential oils from plants. |
| Why is vacuum distillation necessary? | For liquids with very high boiling points or those that decompose when heated to their boiling points under atmospheric pressure, reducing pressure lowers the boiling point. |
| What is an azeotrope, and how does it affect distillation? | An azeotrope is a mixture that has a constant boiling point and cannot be separated by simple or fractional distillation; azeotropic distillation is used to break the azeotrope. |
| How do you improve the purity of the distillate? | By slowing down the distillation rate, using a fractionating column, maintaining a constant temperature, and optimizing the reflux ratio. |
| What are the key components of a basic distillation setup? | Still or retort, heat source, condenser, receiver, and thermometer. |
| How is distillation used in the petroleum refining industry? | To separate crude oil into various components such as gasoline, kerosene, diesel, and lubricating oils through fractional distillation. |
| What safety precautions should be taken during distillation? | Wear safety goggles, use a fume hood, avoid overheating, never distill to dryness, and ensure proper disposal of chemical waste. |
| How can AI be used in distillation processes? | For process monitoring and control, process modeling and simulation, fault detection and diagnosis, predictive maintenance, and process optimization. |
Do you still have questions about distillation or any other topic? At WHAT.EDU.VN, we’re committed to providing clear, accurate answers to all your questions. Don’t hesitate to ask us anything! Our team of experts is ready to help you understand even the most complex subjects. Contact us today at 888 Question City Plaza, Seattle, WA 98101, United States, or reach out via Whatsapp at +1 (206) 555-7890. You can also visit our website at WHAT.EDU.VN for more information and to submit your questions. Get the answers you need quickly and easily with what.edu.vn.