What Is Soluble? Solubility is the property of a substance (solute) to dissolve in a solvent. At WHAT.EDU.VN, we explain this concept simply and offer a free platform to ask all your questions. Explore factors affecting solubility, including temperature, pressure, and intermolecular forces, plus get tips for dissolving substances.
1. Understanding the Basics: What is Solubility?
Solubility is a crucial concept in chemistry and everyday life. It refers to the ability of a substance, known as a solute, to dissolve in another substance, known as a solvent, to form a solution. A solution is a homogeneous mixture where the solute is evenly distributed throughout the solvent.
1.1. Defining Key Terms: Solute, Solvent, and Solution
To truly grasp “what is soluble,” we must define these three key terms:
- Solute: The substance that dissolves. It can be a solid, liquid, or gas. Salt and sugar are common examples of solid solutes.
- Solvent: The substance that dissolves the solute. It is typically a liquid but can also be a gas or a solid. Water is often referred to as the “universal solvent” due to its ability to dissolve a wide range of substances.
- Solution: The homogeneous mixture formed when a solute dissolves in a solvent. Saltwater is a classic example of a solution.
1.2. Solubility: A Deeper Dive
Solubility is more than just whether something dissolves; it’s about how much of a solute can dissolve in a given amount of solvent at a specific temperature. It represents the maximum concentration of a solute that can dissolve in a solvent under defined conditions.
Substances are often categorized based on their solubility:
- Soluble: A substance that dissolves readily in a particular solvent (typically, at least 0.1 g of solute dissolves in 100 mL of solvent).
- Sparingly Soluble (or Slightly Soluble): A substance that dissolves only to a limited extent (less than 0.1 g of solute dissolves in 100 mL of solvent).
- Insoluble: A substance that does not dissolve to a noticeable extent in a particular solvent.
1.3. Expressing Solubility
Solubility can be expressed in various units:
- Grams per liter (g/L): This indicates the mass of solute that dissolves in one liter of saturated solution. For example, the solubility of a substance in water might be expressed as 15 g/L at 20°C.
- Moles per liter (mol/L) or Molarity (M): This represents the number of moles of solute that dissolve in one liter of saturated solution. For example, a molar solubility could be 0.25 mol/L at 25°C.
- Percentage (%): This indicates the mass or volume of solute per 100 units of solvent.
2. Factors Affecting Solubility: What Influences How Much Dissolves?
Several factors influence the solubility of a solute in a solvent. Understanding these factors is crucial for predicting and controlling the dissolution process.
2.1. The Role of Temperature
Temperature plays a significant role in solubility, but its effect differs depending on whether the solute is a solid, liquid, or gas.
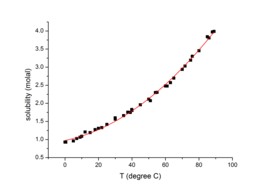
- Solids in Liquids: Generally, the solubility of solid solutes in liquid solvents increases with increasing temperature. This is because higher temperatures provide more energy to break the bonds holding the solute molecules together in the solid state, allowing them to disperse more easily among the solvent molecules.
- Gases in Liquids: The solubility of gases in liquid solvents typically decreases with increasing temperature. As the temperature rises, gas molecules have more kinetic energy and are more likely to escape from the liquid phase. Think of carbonated beverages losing their fizz as they warm up.
2.2. Intermolecular Forces and “Like Dissolves Like”
The strength and type of intermolecular forces between solute and solvent molecules are critical determinants of solubility. The general rule of thumb is “like dissolves like,” meaning that:
- Polar solvents tend to dissolve polar solutes.
- Non-polar solvents tend to dissolve non-polar solutes.
Water, a polar solvent, readily dissolves polar substances like sugar and salt. Oil, a non-polar solvent, dissolves non-polar substances like grease and wax.
The stronger the attractive forces between solute and solvent molecules compared to the attractive forces within the solute and solvent alone, the greater the solubility.
2.3. The Impact of Pressure
Pressure primarily affects the solubility of gases in liquids.
- Gases in Liquids: Henry’s Law states that the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the liquid. This means that increasing the pressure of a gas above a liquid will increase the amount of that gas that dissolves in the liquid. Conversely, decreasing the pressure will decrease the solubility. Carbonated beverages are a perfect example – they are bottled under high pressure to dissolve more carbon dioxide gas.
2.4. Other Factors Influencing Solubility
Besides temperature, intermolecular forces, and pressure, other factors can influence solubility:
- Molecular Size: Larger molecules tend to be less soluble than smaller molecules due to increased intermolecular forces within the solute.
- Crystal Structure: The crystal structure of a solid solute can affect its solubility.
- Presence of Other Solutes: The presence of other solutes in the solvent can sometimes increase or decrease the solubility of a particular solute. This is known as the common ion effect.
3. Solubility of Solids in Liquids: A Closer Examination
The solubility of solids in liquids is a fundamental concept in chemistry with wide-ranging applications.
3.1. The Dissolution Process
When a solid solute is added to a liquid solvent, the process of dissolution begins. Solute molecules at the surface of the solid break away and disperse into the solvent. This process is driven by the attractive forces between the solute and solvent molecules.
3.2. Dynamic Equilibrium: Saturated, Unsaturated, and Supersaturated Solutions
As the dissolution process continues, the concentration of solute in the solution increases. Eventually, a point is reached where the rate of dissolution is equal to the rate of precipitation (the reverse process where solute molecules come out of solution and re-form the solid). This state is called dynamic equilibrium.
At dynamic equilibrium, the solution is said to be saturated. A saturated solution contains the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature.
- Unsaturated Solution: A solution that contains less solute than the maximum amount that can dissolve is called an unsaturated solution. More solute can be added to an unsaturated solution, and it will dissolve.
- Supersaturated Solution: Under specific conditions, it is possible to create a supersaturated solution, which contains more solute than a saturated solution. These solutions are unstable, and the excess solute will typically precipitate out of the solution if disturbed, such as by adding a seed crystal.
3.3. Polarity
Only polar solutes tend to dissolve in the polar solvent and non-polar solvents dissolve only non-polar solutes because of the varying intermolecular forces in a substance
4. Applications of Solubility: Why Is It Important?
Solubility is a crucial concept with numerous applications in various fields.
4.1. Pharmaceutical Industry
Solubility plays a vital role in drug development and delivery. The solubility of a drug affects its absorption, distribution, metabolism, and excretion (ADME) in the body. Poorly soluble drugs may have low bioavailability, meaning that a small amount of the drug reaches the target site.
4.2. Environmental Science
Solubility is essential for understanding the transport and fate of pollutants in the environment. The solubility of a pollutant determines how easily it can dissolve in water and spread through soil and water systems.
4.3. Food Science
Solubility is crucial in food processing and preparation. For example, the solubility of sugar in water affects the sweetness and texture of food products.
4.4. Chemical Reactions
Solubility is fundamental to many chemical reactions. Reactions often occur in solution, and the solubility of reactants and products can significantly affect the reaction rate and equilibrium.
5. Frequently Asked Questions About Solubility
To further clarify the concept of “what is soluble,” here are some frequently asked questions.
| Question | Answer |
|---|---|
| What is the difference between solubility and rate of dissolution? | Solubility refers to the amount of solute that can dissolve in a solvent at equilibrium, while the rate of dissolution refers to how quickly a solute dissolves. |
| Does stirring affect solubility? | Stirring does not affect solubility itself, but it increases the rate of dissolution by bringing fresh solvent into contact with the solute. |
| What are some examples of insoluble substances? | Examples of insoluble substances include sand in water, oil in water, and gold in most common solvents. |
| How can I increase the solubility of a solid in a liquid? | You can often increase the solubility of a solid in a liquid by increasing the temperature or by choosing a solvent that has similar intermolecular forces to the solute. |
| What is a saturated solution? | A saturated solution contains the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature. At this point, the rate of dissolution is equal to the rate of precipitation, creating a dynamic equilibrium. |
| Why is water called the “universal solvent”? | Water is called the “universal solvent” because it can dissolve a wide variety of substances, particularly polar and ionic compounds. Its polarity and ability to form hydrogen bonds make it an excellent solvent for many solutes. |
| How does pressure affect the solubility of solids and liquids? | Pressure has a negligible effect on the solubility of solids and liquids. It primarily affects the solubility of gases in liquids. |
| What is Henry’s Law? | Henry’s Law states that the solubility of a gas in a liquid is directly proportional to the partial pressure of the gas above the liquid. This means that increasing the pressure of a gas above a liquid will increase the amount of that gas that dissolves in the liquid. |
| What is the “like dissolves like” principle? | The “like dissolves like” principle states that polar solvents tend to dissolve polar solutes, and non-polar solvents tend to dissolve non-polar solutes. This is because substances with similar intermolecular forces are more likely to mix and form a solution. |
| How is solubility used in everyday life? | Solubility is used in many aspects of everyday life, including cooking (dissolving sugar in water), cleaning (dissolving soap in water), and taking medications (dissolving drugs in the body). |
| What is molar solubility? | Molar solubility is the number of moles of solute per one liter of saturated solution. For example, 0.115 mol/L at 25 oC. |
| What happens when no more solute can be dissolved into a solvent? | When no more solute can be dissolved into a solvent the solution is considered saturated. This is when molarity (M) and solubility become directly related. The molarity of the solute is equal to its solubility. When no more solute can be added to the solution then the solution is unsaturated. |


6. Need More Answers? Ask WHAT.EDU.VN!
Understanding “what is soluble” and the factors that influence solubility can seem complex. If you have further questions or need clarification on any aspect of this topic, don’t hesitate to ask!
At WHAT.EDU.VN, we provide a free platform for you to ask any question and receive helpful answers from experts and community members. Whether you’re a student, a professional, or simply curious about the world around you, we’re here to help you find the information you need.
Here’s how WHAT.EDU.VN can help you:
- Ask any question, no matter how simple or complex.
- Get answers quickly and easily.
- Access a wealth of information and knowledge.
- Connect with a community of learners and experts.
Don’t struggle with your questions alone. Visit WHAT.EDU.VN today and get the answers you’re looking for!
Our contact information:
- Address: 888 Question City Plaza, Seattle, WA 98101, United States
- WhatsApp: +1 (206) 555-7890
- Website: what.edu.vn
We are always available and willing to answer all of your questions.
