What Is The Heaviest Element? Oganesson, the heaviest element currently known, possesses an intriguing atomic mass. At WHAT.EDU.VN, we’re committed to clarifying complex scientific concepts, providing easy-to-understand answers. Delve into the characteristics of superheavy elements and understand their exotic properties.
1. Understanding the Heaviest Element: Oganesson
Oganesson, with an atomic number of 118, reigns supreme as the heaviest element on the periodic table. This synthetic element, symbolized as Og, carries an approximate atomic mass of 300. Named in honor of the pioneering Russian physicist Yuri Oganessian, it has captured the fascination of scientists worldwide due to its unique properties and behaviors.
-
Discovery and Synthesis: Oganesson’s creation is no small feat. It requires specialized facilities and sophisticated techniques. First synthesized in 2002 by a joint team of Russian and American scientists at the Joint Institute for Nuclear Research (JINR) in Dubna, Russia, it’s produced by bombarding Californium-249 atoms with Calcium-48 ions.
-
Extreme Instability: Oganesson atoms are fleeting, existing for less than a millisecond before decaying. This extreme instability makes experimental study challenging, pushing scientists to rely on theoretical models and computational predictions to understand its characteristics.
-
Position on the Periodic Table: Oganesson resides in Group 18 (the noble gases) and Period 7 of the periodic table. It’s located right after Tennessine (Ts), which further emphasizes its position as the endpoint of the known elements.
2. Key Properties of Oganesson
Because of its fleeting existence, many of Oganesson’s properties remain theoretical. However, sophisticated calculations and indirect measurements offer us insight into what this superheavy element might be like.
-
Electronic Structure: Oganesson’s electronic configuration is predicted to be [Rn] 5f14 6d10 7s2 7p6. However, relativistic effects, stemming from the high speeds of electrons in such heavy atoms, are expected to play a significant role in its electronic structure, making it deviate from what simple extrapolation would suggest.
-
Relativistic Effects: In elements as massive as Oganesson, the inner electrons move at speeds approaching the speed of light. This causes their mass to increase, leading to changes in their orbital shapes and energy levels. These relativistic effects dramatically influence chemical properties, making Oganesson behave quite differently from lighter noble gases.
-
Predicted Chemical Behavior: Based on relativistic calculations, Oganesson is expected to exhibit unusual chemical behavior compared to lighter noble gases. While noble gases are known for their inertness, Oganesson might be more reactive. It’s suggested it could form chemical compounds, which is quite contrary to the non-reactive nature of other noble gases.
3. The Enigmatic Electron Cloud
One of the most intriguing predictions about Oganesson involves its electron configuration. Unlike lighter elements, where electrons reside in discrete, well-defined shells around the nucleus, Oganesson’s electrons are hypothesized to form a “nebulous blob.”
-
Classical vs. Relativistic Models: Classical physics suggests that Oganesson’s electrons should arrange themselves in shells, similar to Xenon and Radon. However, when Einstein’s theory of special relativity is factored in, a different picture emerges. The high speed of electrons in Oganesson cause relativistic effects, leading to the electrons appearing as a spread-out, indistinct blob.
-
Implications of the Blob: This unusual electron arrangement has significant implications for how Oganesson interacts with other atoms. Instead of clear-cut electron sharing or transfer, the nebulous electron cloud could lead to unique bonding behaviors, potentially enabling Oganesson to form compounds that defy conventional chemical intuition.
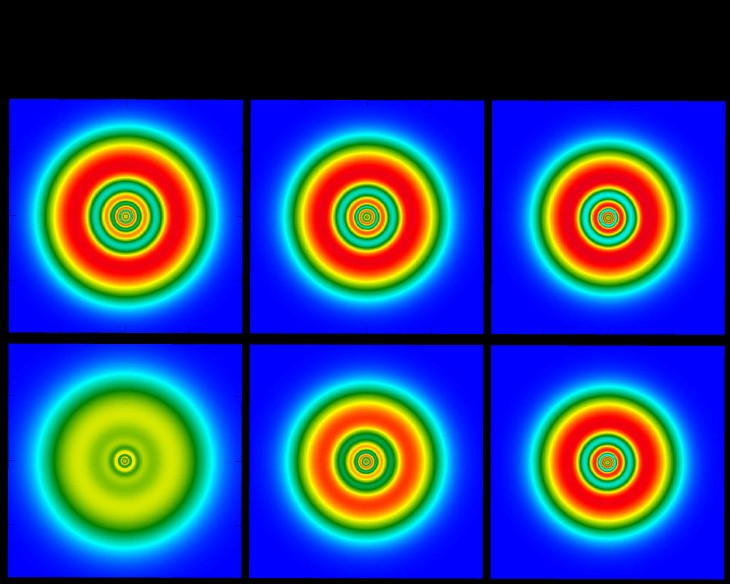 Oganesson Electron Shells
Oganesson Electron Shells
4. Reactivity of a Noble Gas?
The noble gases are renowned for their reluctance to engage in chemical reactions. However, Oganesson might break this trend. Theoretical predictions suggest that it could be chemically reactive, standing in stark contrast to its lighter noble gas counterparts.
-
Electron Affinity and Ionization Energy: Electron affinity measures the energy change when an electron is added to a neutral atom, while ionization energy measures the energy required to remove an electron. For typical noble gases, ionization energies are very high, and electron affinities are very low, indicating their stability and resistance to forming chemical bonds. Oganesson, due to relativistic effects, could have a lower ionization energy and a higher electron affinity than expected, making it more prone to chemical interactions.
-
Potential Compound Formation: It’s hypothesized that Oganesson could form compounds with highly electronegative elements like fluorine or oxygen. These compounds might be stabilized by relativistic effects and could exhibit unusual bonding characteristics.
5. Is Oganesson a Solid at Room Temperature?
Gases are expected to be gaseous at room temperature. However, intriguing predictions propose that Oganesson could buck this trend and exist as a solid under normal conditions.
-
Interatomic Interactions: The physical state of a substance depends on the strength of the interactions between its atoms or molecules. In noble gases, these interactions are weak, leading to their gaseous state. However, in Oganesson, relativistic effects could enhance these interactions, leading to stronger attraction between atoms.
-
Formation of Clusters: If the interatomic forces in Oganesson are strong enough, atoms could clump together to form clusters. At room temperature, these clusters could potentially form a solid-state, making Oganesson a unique noble gas that transitions to solid form under standard conditions.
6. The Bubble Nucleus Hypothesis
Within the nucleus of an atom, protons, which carry positive charges, repel each other. Ordinarily, the strong nuclear force overcomes this repulsion, binding the protons and neutrons together. However, in Oganesson, the sheer number of protons (118) might create a unique phenomenon – a “bubble nucleus.”
-
Proton Distribution: A bubble nucleus implies a spatial distribution of protons where the center of the nucleus is relatively depleted of protons. This can occur when the repulsive forces between protons become significant enough to push them away from the center.
-
Implications for Nuclear Stability: The formation of a bubble nucleus could impact the overall stability of the Oganesson nucleus. It might influence the element’s decay modes and half-life. Experimental evidence for bubble nuclei has been found in lighter elements, providing some support for this hypothesis.
7. Neutron Behavior in Superheavy Elements
The behavior of neutrons in superheavy elements like Oganesson is another area of active research. Neutrons, being neutral particles, don’t experience the same electrostatic repulsion as protons. However, their distribution and arrangement within the nucleus contribute to the overall stability and properties of the element.
-
Neutron Shells: In some heavy elements, neutrons are arranged in well-defined shells within the nucleus. However, predictions suggest that in Oganesson, these neutron shells might be less distinct, with neutrons mingling more freely.
-
Impact on Nuclear Structure: The way neutrons are arranged in the nucleus can influence the shape and stability of the nucleus. A more uniform distribution of neutrons might lead to a more spherical nucleus, while distinct neutron shells could result in a deformed nucleus.
8. Experimental Challenges and Future Directions
Studying Oganesson poses formidable experimental challenges due to its fleeting existence and the difficulty in producing it in sufficient quantities. However, scientists are pushing the boundaries of technology to explore this fascinating element.
-
Superheavy Element Factories: New facilities are being constructed around the world to synthesize and study superheavy elements. These facilities utilize advanced particle accelerators and detection systems to produce and characterize these exotic atoms.
-
Indirect Measurements: Since direct measurements on Oganesson are challenging, scientists often rely on indirect measurements. For example, they can study the decay products of Oganesson to infer its properties. They can also perform sophisticated calculations that predict its behavior based on the fundamental laws of physics.
-
The Role of Theory: Theoretical calculations play a crucial role in understanding Oganesson. Scientists use powerful computers and advanced algorithms to model the electronic structure and nuclear properties of Oganesson. These calculations provide valuable insights that guide experiments and help interpret experimental results.
9. Oganesson in Popular Culture and Education
Even though it is an obscure element, Oganesson’s unique status as the heaviest element has earned it a place in popular culture and education.
-
Periodic Table Updates: The inclusion of Oganesson in the periodic table highlights the dynamic nature of science. It shows that the periodic table is not a static entity but rather a constantly evolving representation of our understanding of the elements.
-
Inspiring Future Scientists: Oganesson’s story, from its synthesis to its theoretical properties, inspires young scientists to explore the unknown and push the boundaries of knowledge. It demonstrates the power of scientific inquiry and the thrill of discovery.
10. FAQ: Unveiling More About The Heaviest Element
| Question | Answer |
|---|---|
| What is Oganesson’s symbol? | Og |
| Who is Oganesson named after? | Yuri Oganessian, a Russian physicist. |
| How many protons does Oganesson have? | 118 |
| Is Oganesson naturally occurring? | No, it is a synthetic element. |
| What is the predicted state of Oganesson? | Potentially a solid at room temperature. |
| Where was Oganesson first synthesized? | Joint Institute for Nuclear Research (JINR) in Dubna, Russia. |
| Why is Oganesson difficult to study? | It’s extremely unstable and exists for only a fraction of a second. |
| What are relativistic effects? | Changes in the behavior of electrons due to their high speeds in heavy atoms. |
| Could Oganesson form chemical compounds? | Theoretical predictions suggest it could, unlike other noble gases. |
| What makes Oganesson’s nucleus unique? | The potential formation of a “bubble nucleus” due to proton repulsion. |
Navigating the complexities of science can be daunting. At WHAT.EDU.VN, we believe everyone deserves access to clear, concise, and reliable information. Our platform offers a unique opportunity to ask any question and receive insightful answers from knowledgeable experts, completely free of charge. Don’t let your curiosity be stifled. Visit what.edu.vn today at 888 Question City Plaza, Seattle, WA 98101, United States, or reach us on Whatsapp at +1 (206) 555-7890 to get your questions answered.