What Is The Periodic Table? It’s a cornerstone of chemistry and understanding the building blocks of our world. At WHAT.EDU.VN, we provide accessible explanations and expert insights, making complex topics simple. Discover the arrangement, elements, and periodic trends.
Table of Contents
- What is the Periodic Table and Its Significance?
- Who Invented the Periodic Table?
- How is the Periodic Table Organized?
- Decoding Element Symbols on the Periodic Table
- Metals, Nonmetals, and Metalloids: A Periodic Table Overview
- Understanding the Periodic Trends
- Groups of the Periodic Table Explained
- Periods of the Periodic Table: What They Reveal
- Applications of the Periodic Table in Everyday Life
- The Periodic Table’s Role in Predicting Chemical Properties
- How the Periodic Table Helps in Balancing Chemical Equations
- The Periodic Table’s Impact on Discovering New Elements
- Periodic Table and the Electronic Configuration
- Understanding Oxidation States Using the Periodic Table
- The Periodic Table’s Contribution to Material Science
- Periodic Table and Environmental Science
- How Periodic Table Helps in Drug Discovery
- The Periodic Table’s Influence on Nanotechnology
- Periodic Table and Nuclear Chemistry
- The Periodic Table’s Future: What’s Next?
- Periodic Table FAQs: Answered by Experts
1. What is the Periodic Table and Its Significance?
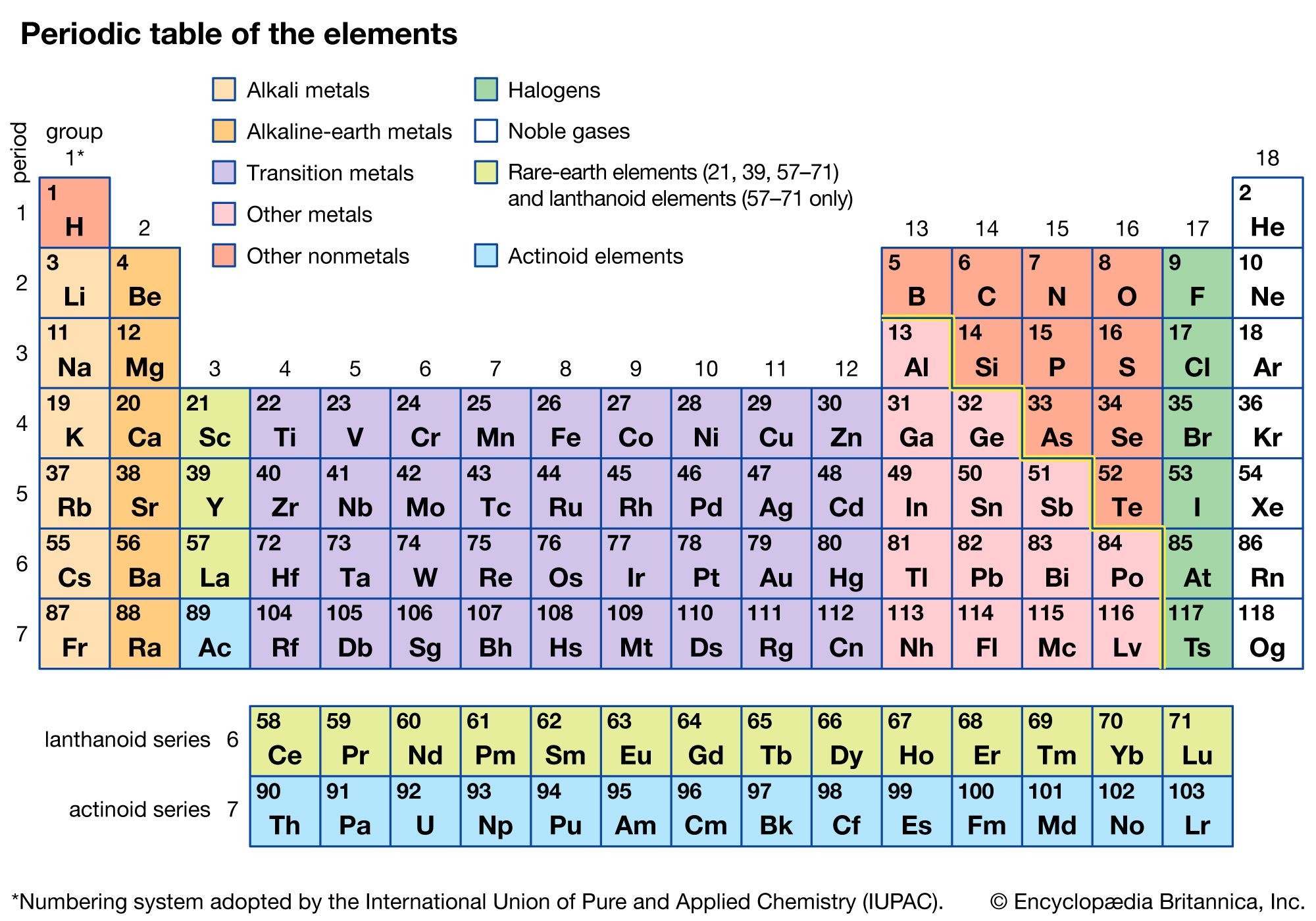
The periodic table is a tabular arrangement of chemical elements, ordered by their atomic number, electron configuration, and recurring chemical properties. It serves as an indispensable tool in chemistry, offering a structured framework for understanding the properties and behaviors of elements. It’s more than just a chart; it’s a map of the chemical world. Think of it as a cheat sheet that lets you easily predict how elements might behave in various reactions.
The periodic table’s significance lies in its ability to:
- Organize Knowledge: It consolidates a vast amount of information about elements in an accessible format.
- Predict Properties: It enables scientists to predict the properties of elements based on their position in the table.
- Facilitate Research: It guides research by suggesting possible chemical reactions and compound formations.
- Educational Tool: It is crucial for teaching and learning chemistry at all levels.
The periodic table organizes elements by atomic number and recurring chemical properties.
Is there something you’ve always wanted to know about the periodic table, but haven’t found a clear answer? Visit WHAT.EDU.VN and ask your question for free!
2. Who Invented the Periodic Table?
The creation of the periodic table is primarily credited to Dmitri Mendeleev, a Russian chemist. In 1869, Mendeleev published his periodic table, which arranged elements by atomic weight and grouped them based on similar properties. This initial table had gaps, which Mendeleev boldly predicted would be filled by then-undiscovered elements. His predictions proved accurate, solidifying the table’s credibility and utility.
However, Mendeleev was not the only scientist working on this problem. Julius Lothar Meyer, a German chemist, also developed a similar table around the same time. While Meyer’s table was also based on atomic weights and recurring properties, Mendeleev is generally given more credit for his accurate predictions.
Dmitri Mendeleev is credited with creating the first periodic table.
Have you ever wondered how scientists predict the properties of elements? Ask your question at WHAT.EDU.VN and get expert insights without any cost!
3. How is the Periodic Table Organized?
The periodic table is organized in a specific manner to reflect the periodic law, which states that the properties of elements are periodic functions of their atomic numbers. This organization is based on:
- Atomic Number: Elements are arranged in increasing order of their atomic number (the number of protons in an atom).
- Electron Configuration: The arrangement reflects the electron configuration of elements, which determines their chemical properties.
- Groups (Columns): Vertical columns, known as groups or families, contain elements with similar chemical properties due to having the same number of valence electrons.
- Periods (Rows): Horizontal rows, known as periods, represent elements with the same number of electron shells.
- Blocks: The table is divided into blocks (s, p, d, and f) based on the type of subshell being filled by the element’s outermost electrons.
The periodic table is organized into blocks (s, p, d, f) based on electron configuration.
If you are confused about electron configuration, WHAT.EDU.VN is here to help. Ask your questions and receive clear and concise answers from experts.
4. Decoding Element Symbols on the Periodic Table
Each element on the periodic table is represented by a unique symbol, typically one or two letters derived from its name (often Latin). For example:
- H stands for Hydrogen
- O stands for Oxygen
- Na stands for Sodium (from the Latin “Natrium”)
- Fe stands for Iron (from the Latin “Ferrum”)
In addition to the symbol, each element’s cell on the periodic table usually includes its atomic number and atomic mass. The atomic number is the number of protons in the nucleus, while the atomic mass is the average mass of an atom of that element, usually expressed in atomic mass units (amu).
Each element cell typically contains the element symbol, atomic number, and atomic mass.
Do you want to know more about the origin of element symbols and names? WHAT.EDU.VN provides access to a community of experts who can answer all your questions for free.
5. Metals, Nonmetals, and Metalloids: A Periodic Table Overview
Elements in the periodic table can be broadly classified into three categories: metals, nonmetals, and metalloids.
- Metals: Generally located on the left side of the table, metals are typically shiny, good conductors of electricity and heat, and malleable (can be hammered into sheets) and ductile (can be drawn into wires). Examples include iron (Fe), gold (Au), and copper (Cu).
- Nonmetals: Found on the right side of the table, nonmetals generally lack metallic properties. They are often poor conductors of heat and electricity and can be solids, liquids, or gases at room temperature. Examples include oxygen (O), sulfur (S), and chlorine (Cl).
- Metalloids: Located along the staircase line that separates metals and nonmetals, metalloids (also known as semi-metals) have properties intermediate between metals and nonmetals. They are often semiconductors, making them useful in electronics. Examples include silicon (Si) and germanium (Ge).
Metals, nonmetals, and metalloids are categorized on the periodic table based on their properties.
Are you curious about the specific properties of metals, nonmetals, or metalloids? WHAT.EDU.VN offers expert answers to all your chemistry questions at no cost.
6. Understanding the Periodic Trends
Periodic trends are specific patterns in the properties of elements that vary predictably across the periodic table. Understanding these trends is crucial for predicting how elements will behave. The main periodic trends include:
- Atomic Radius: Generally decreases from left to right across a period and increases down a group.
- Ionization Energy: Generally increases from left to right across a period and decreases down a group.
- Electronegativity: Generally increases from left to right across a period and decreases down a group.
- Electron Affinity: Generally increases from left to right across a period and decreases down a group (though there are exceptions).
- Metallic Character: Decreases from left to right across a period and increases down a group.
Periodic trends show predictable variations in element properties across the table.
Confused about periodic trends? Don’t worry! Ask your questions on WHAT.EDU.VN and get detailed explanations from chemistry experts.
7. Groups of the Periodic Table Explained
The vertical columns in the periodic table are called groups or families. Elements within the same group share similar chemical properties because they have the same number of valence electrons. Some important groups include:
- Alkali Metals (Group 1): Highly reactive metals that readily lose one electron to form positive ions.
- Alkaline Earth Metals (Group 2): Reactive metals that lose two electrons to form positive ions.
- Transition Metals (Groups 3-12): Metals with variable valencies and the ability to form colorful compounds.
- Halogens (Group 17): Highly reactive nonmetals that readily gain one electron to form negative ions.
- Noble Gases (Group 18): Inert gases with complete valence shells, making them very stable.
Elements in the same group share similar chemical properties.
Want to know more about the unique properties of each group? Post your questions on WHAT.EDU.VN and get expert answers for free.
8. Periods of the Periodic Table: What They Reveal
The horizontal rows in the periodic table are called periods. Elements within the same period have the same number of electron shells. As you move from left to right across a period, the atomic number increases, and the elements exhibit a gradual change in properties from metallic to nonmetallic.
- Period 1: Contains only hydrogen (H) and helium (He).
- Period 2: Contains lithium (Li) to neon (Ne).
- Period 3: Contains sodium (Na) to argon (Ar).
- Period 4: Contains potassium (K) to krypton (Kr).
- Period 5: Contains rubidium (Rb) to xenon (Xe).
- Period 6: Contains cesium (Cs) to radon (Rn), including lanthanides.
- Period 7: Contains francium (Fr) to oganesson (Og), including actinides.
Each period represents elements with the same number of electron shells.
Interested in the trends within each period? Ask your specific questions on WHAT.EDU.VN and receive detailed explanations from knowledgeable experts.
9. Applications of the Periodic Table in Everyday Life
The periodic table isn’t just an abstract chart for chemists; it has numerous practical applications in everyday life:
- Electronics: Silicon (Si) and germanium (Ge) are used in semiconductors for electronic devices.
- Construction: Iron (Fe) is a primary component of steel, used in building infrastructure.
- Medicine: Iodine (I) is used as a disinfectant, and various elements are used in medical imaging.
- Household Items: Aluminum (Al) is used in cookware, and chlorine (Cl) is used in water purification.
- Agriculture: Nitrogen (N), phosphorus (P), and potassium (K) are essential nutrients in fertilizers.
Elements have diverse applications in electronics, construction, medicine, and more.
Curious about how specific elements impact your daily life? Ask your questions on WHAT.EDU.VN and get clear, informative answers from our team of experts.
10. The Periodic Table’s Role in Predicting Chemical Properties
The periodic table is instrumental in predicting chemical properties of elements and their compounds. By understanding the trends in electronegativity, ionization energy, and atomic size, chemists can anticipate how elements will react with each other.
- Reactivity: Elements in the same group tend to have similar reactivity. For example, alkali metals (Group 1) are highly reactive and readily form compounds with halogens (Group 17).
- Bonding: Electronegativity differences between elements can predict the type of bond that will form (ionic, covalent, or metallic).
- Acid-Base Behavior: The position of an element in the periodic table can provide clues about the acidic or basic nature of its oxides and hydroxides.
Periodic trends allow for the prediction of chemical properties and reactivity.
Do you want to know how to predict the outcome of chemical reactions using the periodic table? Get your answers quickly and easily by asking on WHAT.EDU.VN.
11. How the Periodic Table Helps in Balancing Chemical Equations
Balancing chemical equations is a fundamental skill in chemistry, and the periodic table can assist in this process. By knowing the common oxidation states of elements, you can predict the formulas of compounds and ensure that the number of atoms of each element is the same on both sides of the equation.
- Identifying Ions: The periodic table helps identify the common ions formed by elements. For example, Group 1 elements typically form +1 ions, while Group 17 elements typically form -1 ions.
- Predicting Compound Formulas: Knowing the charges of ions allows you to predict the formulas of ionic compounds. For example, sodium (Na+) and chlorine (Cl-) combine to form NaCl.
- Balancing Redox Reactions: The periodic table helps in assigning oxidation numbers, which is crucial for balancing redox reactions.
The periodic table is a tool for balancing chemical equations.
Having trouble balancing chemical equations? Post your equation on WHAT.EDU.VN, and let our experts guide you through the balancing process step by step. It’s free and fast!
12. The Periodic Table’s Impact on Discovering New Elements
The periodic table has played a crucial role in the discovery of new elements. Mendeleev’s original table had gaps, which he correctly predicted would be filled by elements that had not yet been discovered. The properties of these undiscovered elements were also predicted based on their expected position in the table.
- Predicting Properties: The periodic table provides a framework for predicting the properties of new elements, guiding experimental efforts to synthesize and characterize them.
- Filling Gaps: The discovery of elements like gallium (Ga), germanium (Ge), and scandium (Sc) validated Mendeleev’s predictions and cemented the periodic table’s importance.
- Synthesizing Transuranic Elements: The periodic table has guided the synthesis of transuranic elements (elements with atomic numbers greater than 92) in nuclear reactors and particle accelerators.
The periodic table guides the discovery and synthesis of new elements.
What does it take to discover a new element? Ask your questions on WHAT.EDU.VN and get insights into the cutting-edge research in this field.
13. Periodic Table and the Electronic Configuration
The arrangement of elements in the periodic table is directly related to their electronic configuration. The electronic configuration describes how electrons are arranged within an atom, and it determines the chemical properties of the element.
- Valence Electrons: Elements in the same group have the same number of valence electrons (electrons in the outermost shell), which explains their similar chemical behavior.
- Blocks and Subshells: The s, p, d, and f blocks correspond to the filling of the s, p, d, and f subshells, respectively.
- Predicting Configurations: The periodic table can be used to predict the electronic configuration of an element. For example, potassium (K) is in Group 1 and Period 4, so its electronic configuration ends in 4s1.
The periodic table reflects the electronic configuration of elements.
Do you need help understanding electronic configurations? Ask your questions on WHAT.EDU.VN and receive clear explanations and examples from our chemistry experts.
14. Understanding Oxidation States Using the Periodic Table
Oxidation states, also known as oxidation numbers, represent the degree of oxidation of an atom in a chemical compound. The periodic table can help predict the common oxidation states of elements.
- Group Numbers: Elements in Group 1 typically have an oxidation state of +1, while elements in Group 2 have an oxidation state of +2.
- Halogens: Halogens (Group 17) typically have an oxidation state of -1.
- Transition Metals: Transition metals can have multiple oxidation states, which contributes to their diverse chemistry.
:max_bytes(150000):strip_icc():format(webp)/138744681-56a0499b5f9b58eba4b0c18a.jpg)
The periodic table helps predict common oxidation states.
Want to know more about oxidation states and how they influence chemical reactions? Ask your questions on WHAT.EDU.VN and get detailed answers from experienced chemists.
15. The Periodic Table’s Contribution to Material Science
The periodic table is essential in material science, guiding the development of new materials with specific properties.
- Alloys: By combining different metals, alloys with enhanced strength, corrosion resistance, or other desired properties can be created.
- Semiconductors: Metalloids like silicon and germanium are used in semiconductors, which are crucial for electronic devices.
- Polymers: The properties of polymers can be tailored by incorporating different elements into the polymer structure.
- Ceramics: The periodic table helps in selecting elements to create ceramics with high-temperature resistance and hardness.
The periodic table guides the development of new materials.
Interested in the latest advancements in material science? Ask your questions on WHAT.EDU.VN and stay updated with insights from experts in the field.
16. Periodic Table and Environmental Science
The periodic table is relevant to environmental science, helping us understand the distribution and behavior of elements in the environment.
- Pollutants: Identifying and tracking pollutants like heavy metals (e.g., mercury, lead, cadmium) using their positions in the periodic table.
- Nutrient Cycles: Understanding the cycles of essential elements like nitrogen, phosphorus, and carbon.
- Remediation: Developing strategies for cleaning up contaminated sites based on the chemical properties of the pollutants.
The periodic table helps understand element distribution and behavior in the environment.
Have environmental questions? Visit WHAT.EDU.VN to connect with experts who can provide insights into environmental challenges and solutions.
17. How Periodic Table Helps in Drug Discovery
The periodic table plays a significant role in drug discovery by helping medicinal chemists design and synthesize new drug molecules.
- Drug Design: Understanding the properties of elements allows chemists to create molecules that interact specifically with biological targets.
- Metal-Based Drugs: Metals like platinum, gold, and silver are used in certain drugs due to their unique chemical properties.
- Pharmacokinetics: The periodic table aids in predicting how drugs will be absorbed, distributed, metabolized, and excreted by the body.
The periodic table aids in designing and synthesizing new drugs.
Do you have questions about drug development and the role of chemistry? Ask on WHAT.EDU.VN and get reliable answers from experts in the pharmaceutical field.
18. The Periodic Table’s Influence on Nanotechnology
Nanotechnology involves manipulating matter at the nanoscale (1-100 nanometers). The periodic table is crucial in this field for selecting elements with specific properties for nanoscale applications.
- Nanomaterials: Creating nanomaterials with unique properties by combining different elements.
- Quantum Dots: Using semiconductor nanocrystals (quantum dots) for applications in displays, solar cells, and bioimaging.
- Catalysis: Employing nanoparticles as catalysts to speed up chemical reactions.
The periodic table aids in selecting elements for nanoscale applications.
Curious about the potential of nanotechnology? Post your questions on WHAT.EDU.VN and get insights into the latest innovations and applications.
19. Periodic Table and Nuclear Chemistry
Nuclear chemistry involves the study of nuclear reactions, radioactivity, and the properties of radioactive isotopes. The periodic table helps in understanding the behavior of radioactive elements.
- Radioactive Decay: Identifying the types of radioactive decay based on the position of the element in the periodic table.
- Nuclear Reactions: Predicting the products of nuclear reactions and understanding the stability of different isotopes.
- Applications: Using radioactive isotopes in medicine, dating, and energy production.
The periodic table helps understand the behavior of radioactive elements.
Want to learn more about nuclear chemistry? Ask your questions on WHAT.EDU.VN and connect with experts who can explain complex topics in an accessible way.
20. The Periodic Table’s Future: What’s Next?
The periodic table continues to evolve as new elements are synthesized and our understanding of chemistry deepens.
- Discovering New Elements: Scientists are still working to synthesize new superheavy elements and add them to the periodic table.
- Refining Properties: As we learn more about existing elements, their properties are refined, and the periodic table is updated.
- Educational Tools: New interactive and digital versions of the periodic table are being developed to enhance learning and accessibility.
The periodic table continues to evolve with new discoveries.
What are the future challenges and opportunities in chemistry? Ask your questions on WHAT.EDU.VN and engage with experts who are shaping the future of the field.
21. Periodic Table FAQs: Answered by Experts
| Question | Answer |
|---|---|
| What is the most abundant element in the Earth’s crust? | Oxygen (O) is the most abundant element in the Earth’s crust, making up about 46.6% of its mass. |
| Which element is essential for all known life? | Carbon (C) is essential for all known life because it forms the backbone of organic molecules, such as proteins, carbohydrates, and nucleic acids. |
| What are the properties of noble gases? | Noble gases (Group 18) are inert, colorless, odorless, and have very low chemical reactivity due to their complete valence shells. They exist as monatomic gases and are used in lighting, insulation, and other applications. |
| Why are transition metals called “transition”? | Transition metals are called “transition” because their electronic configuration involves the filling of d orbitals, leading to variable oxidation states and diverse chemical properties. They form a bridge between the s-block and p-block elements in the periodic table. |
| What is electronegativity and how does it vary? | Electronegativity is the ability of an atom to attract electrons in a chemical bond. It generally increases from left to right across a period and decreases down a group in the periodic table. Fluorine (F) is the most electronegative element. |
| How are synthetic elements made? | Synthetic elements are made through nuclear reactions in particle accelerators or nuclear reactors. These reactions involve bombarding heavy elements with neutrons or other particles, leading to the formation of new elements with higher atomic numbers. |
| What is the importance of lanthanides and actinides? | Lanthanides and actinides (f-block elements) have unique electronic and magnetic properties, making them useful in various applications. Lanthanides are used in magnets, lighting, and catalysts, while actinides are important in nuclear energy and research. |
| How does atomic radius change across the periodic table? | Atomic radius generally decreases from left to right across a period due to increasing nuclear charge and increases down a group as more electron shells are added. |
| What is ionization energy? | Ionization energy is the energy required to remove an electron from a gaseous atom. It generally increases from left to right across a period and decreases down a group in the periodic table. |
| What is the role of the periodic table in education? | The periodic table is a fundamental tool in chemistry education, providing a structured framework for understanding the properties and behaviors of elements. It helps students learn about atomic structure, chemical bonding, and periodic trends, and it serves as a reference for solving chemical problems and conducting experiments. |

Still have questions about the periodic table? Don’t hesitate! At WHAT.EDU.VN, we are committed to providing free and reliable answers to all your questions. Visit our website today and get the knowledge you need!
Address: 888 Question City Plaza, Seattle, WA 98101, United States
WhatsApp: +1 (206) 555-7890
Website: WHAT.EDU.VN
Unlock Your Potential with what.edu.vn: Ask, Learn, and Grow.