What are polymers? Polymers are large molecules made up of repeating smaller units called monomers, playing a vital role in various aspects of our lives. At WHAT.EDU.VN, we’re dedicated to providing clear, accessible information about polymers and other scientific concepts. Discover more about macromolecules, their composition, and significance, and find answers to all your burning questions.
1. Understanding Polymers: A Comprehensive Introduction
Polymers are ubiquitous. They’re found in nature, from the cellulose that makes up plant cell walls to the proteins that build our bodies. They’re also synthesized in labs, creating plastics, rubbers, and countless other materials that shape our modern world. This section delves into the fundamental definition of polymers, their building blocks (monomers), and the wide range of forms they can take.
1.1. Defining Polymers and Macromolecules
A polymer is essentially a long chain molecule composed of repeating units called monomers. Think of it like a string of beads, where each bead is a monomer and the entire string is the polymer. Because of their size, polymers are also known as macromolecules. These giant molecules can have molecular weights ranging from thousands to millions of atomic mass units.
1.2. Monomers: The Building Blocks of Polymers
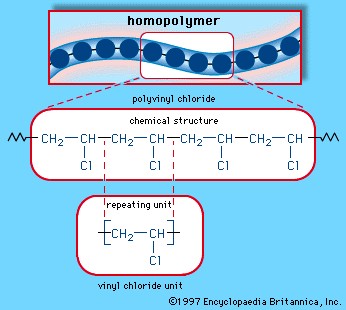
Monomers are the small, repeating units that link together to form polymers. The type of monomer used dictates the properties of the resulting polymer. For example, ethylene monomers link together to form polyethylene, a common plastic used in packaging. Vinyl chloride monomers form polyvinyl chloride (PVC), a rigid plastic used in pipes.
1.3. Homopolymers vs. Copolymers: Different Types of Polymer Structures
Polymers can be classified based on the type of monomers they contain. Homopolymers are made up of only one type of monomer, like polyethylene (just ethylene monomers). Copolymers, on the other hand, are made up of two or more different types of monomers. This allows for a much wider range of properties and applications. For example, styrene-butadiene rubber (SBR), used in tires, is a copolymer of styrene and butadiene.
2. Natural Polymers: Nature’s Own Macromolecules
Nature is a master of polymer synthesis. Natural polymers play essential roles in the structure, function, and life processes of organisms. This section explores some of the most important natural polymers, including proteins, carbohydrates, nucleic acids, and natural rubber.
2.1. Proteins: Polymers of Amino Acids
Proteins are arguably the most versatile and important class of natural polymers. They are polymers of amino acids, linked together by peptide bonds. The sequence of amino acids in a protein determines its unique three-dimensional structure and its specific function. Proteins act as enzymes, catalyzing biochemical reactions; as structural components, providing support for cells and tissues; as hormones, regulating bodily functions; and as antibodies, defending against foreign invaders.
2.2. Carbohydrates: Polymers of Sugars
Carbohydrates are another essential class of natural polymers, providing energy and structural support for living organisms. They are polymers of sugar molecules (monosaccharides), such as glucose, fructose, and galactose. Polysaccharides, such as starch, cellulose, and glycogen, are complex carbohydrates made up of many sugar units linked together. Starch is the primary energy storage molecule in plants, while glycogen serves the same function in animals. Cellulose is the main structural component of plant cell walls, providing rigidity and support.
2.3. Nucleic Acids: Polymers of Nucleotides
Nucleic acids, DNA and RNA, are the information-carrying molecules of life. They are polymers of nucleotides, which are composed of a sugar molecule, a phosphate group, and a nitrogenous base. The sequence of nucleotides in DNA encodes the genetic information that determines the characteristics of an organism. RNA plays a crucial role in protein synthesis, carrying the genetic message from DNA to the ribosomes, where proteins are made.
2.4. Natural Rubber: An Isoprene Polymer
Natural rubber is a polymer of isoprene, a simple hydrocarbon. It is produced by rubber trees and is valued for its elasticity and resilience. Natural rubber is used in a wide range of applications, including tires, hoses, and seals.
3. Synthetic Polymers: Man-Made Marvels
Synthetic polymers are created in laboratories and factories, offering a vast array of materials with tailored properties. This section explores some of the most common and important synthetic polymers, including plastics, rubbers, and fibers.
3.1. Plastics: Versatile and Ubiquitous Polymers
Plastics are synthetic polymers that can be molded into various shapes. They are lightweight, durable, and relatively inexpensive to produce, making them incredibly versatile and ubiquitous in modern life. Different types of plastics have different properties, making them suitable for various applications. Some common examples include:
- Polyethylene (PE): Used in packaging, films, and bottles.
- Polypropylene (PP): Used in containers, fibers, and automotive parts.
- Polyvinyl Chloride (PVC): Used in pipes, flooring, and siding.
- Polystyrene (PS): Used in packaging, insulation, and disposable cups.
- Polyethylene Terephthalate (PET): Used in bottles, fibers, and films.
3.2. Rubbers: Elastic and Resilient Polymers
Synthetic rubbers are polymers that exhibit elasticity and resilience. They can be stretched and deformed but will return to their original shape when the force is removed. Synthetic rubbers are used in a wide range of applications, including tires, seals, and hoses. Some common examples include:
- Styrene-Butadiene Rubber (SBR): Used in tires and footwear.
- Nitrile Rubber (NBR): Used in seals and hoses due to its resistance to oil and chemicals.
- Silicone Rubber: Used in seals, adhesives, and medical implants due to its heat resistance and biocompatibility.
3.3. Fibers: Strong and Flexible Polymers
Synthetic fibers are polymers that can be spun into long, thin strands. They are strong, flexible, and can be woven into fabrics. Synthetic fibers are used in clothing, carpets, and industrial applications. Some common examples include:
- Nylon: Used in clothing, ropes, and carpets.
- Polyester: Used in clothing, bottles, and films.
- Acrylic: Used in clothing, carpets, and upholstery.
4. Polymer Properties: What Makes Them Unique?
The properties of polymers are determined by their chemical composition, molecular weight, and structure. These properties dictate how polymers behave and what applications they are suitable for. This section explores some of the key properties of polymers, including their strength, flexibility, elasticity, and thermal behavior.
4.1. Strength and Flexibility: Balancing Act in Polymers
The strength of a polymer refers to its ability to withstand stress without breaking. Flexibility refers to its ability to bend without breaking. These two properties are often related, as stronger polymers tend to be less flexible, and vice versa. The strength and flexibility of a polymer depend on the intermolecular forces between the polymer chains. Stronger intermolecular forces lead to higher strength but lower flexibility.
4.2. Elasticity: The Ability to Return to Original Shape
Elasticity is the ability of a polymer to return to its original shape after being deformed. This property is particularly important for rubbers and elastomers. Elasticity depends on the ability of the polymer chains to stretch and recoil. Polymers with a high degree of cross-linking tend to have higher elasticity.
4.3. Thermal Behavior: How Polymers Respond to Heat
The thermal behavior of a polymer refers to how it responds to changes in temperature. Some polymers are thermoplastic, meaning they can be melted and reshaped repeatedly. Others are thermosetting, meaning they undergo an irreversible chemical change when heated, forming a rigid, cross-linked structure. Thermoplastics are generally more flexible and easier to process than thermosets.
5. Polymer Synthesis: Building Macromolecules
Polymer synthesis is the process of joining monomers together to form polymers. There are two main types of polymerization: addition polymerization and condensation polymerization. This section explores these two methods in detail.
5.1. Addition Polymerization: Chain Growth
Addition polymerization involves the joining of monomers without the loss of any atoms. This process typically involves a chain reaction, where an initiator molecule starts the polymerization process, and the chain grows rapidly as monomers are added to the growing polymer chain. Polyethylene, polypropylene, and PVC are all synthesized by addition polymerization.
5.2. Condensation Polymerization: Step Growth
Condensation polymerization involves the joining of monomers with the loss of a small molecule, such as water. This process typically occurs in a step-wise fashion, where monomers react with each other to form dimers, trimers, and eventually long polymer chains. Nylon, polyester, and polyurethanes are all synthesized by condensation polymerization.
6. Polymer Applications: Shaping Our World
Polymers are used in a vast array of applications, from packaging and clothing to construction and medicine. Their versatility, durability, and cost-effectiveness make them indispensable materials in modern society. This section explores some of the most important and innovative applications of polymers.
6.1. Packaging: Protecting and Preserving Goods
Polymers are widely used in packaging to protect and preserve goods during transport and storage. Plastic films, containers, and foams are used to package food, beverages, electronics, and countless other products. Polymers provide a barrier against moisture, oxygen, and other contaminants, extending the shelf life of products.
6.2. Clothing: Comfort, Style, and Performance
Synthetic fibers, such as nylon, polyester, and acrylic, are used extensively in clothing to provide comfort, style, and performance. These fibers are strong, flexible, and can be woven into fabrics that are durable, wrinkle-resistant, and easy to care for. Synthetic fibers are also used in sportswear to provide moisture-wicking and breathable properties.
6.3. Construction: Building Strong and Durable Structures
Polymers are used in construction to build strong and durable structures. PVC pipes, roofing membranes, and insulation materials are all made from polymers. Polymers provide resistance to corrosion, weather, and pests, extending the lifespan of buildings and infrastructure.
6.4. Medicine: Improving Healthcare and Saving Lives
Polymers are used in medicine to improve healthcare and save lives. Medical implants, drug delivery systems, and wound dressings are all made from polymers. Polymers are biocompatible, meaning they do not cause adverse reactions in the body. They can also be designed to release drugs slowly over time, providing sustained therapeutic effects.
7. Polymer Recycling: Addressing Environmental Concerns
The widespread use of polymers has led to environmental concerns about plastic waste and pollution. Polymer recycling is the process of recovering and reprocessing waste polymers into new products. Recycling reduces the amount of plastic waste that ends up in landfills and oceans, conserving resources and reducing pollution.
7.1. Types of Polymer Recycling
There are several types of polymer recycling, including:
- Mechanical Recycling: Involves melting and reshaping waste polymers into new products.
- Chemical Recycling: Involves breaking down waste polymers into their constituent monomers, which can then be used to synthesize new polymers.
- Energy Recovery: Involves burning waste polymers to generate energy.
7.2. Challenges and Opportunities in Polymer Recycling
Polymer recycling faces several challenges, including the difficulty of separating different types of polymers, the presence of contaminants in waste polymers, and the lack of infrastructure for collecting and processing waste polymers. However, there are also significant opportunities to improve polymer recycling, including the development of new recycling technologies, the design of polymers that are easier to recycle, and the implementation of policies that promote recycling.
8. The Future of Polymers: Innovation and Sustainability
The field of polymer science is constantly evolving, with new discoveries and innovations emerging all the time. The future of polymers is focused on developing sustainable and high-performance materials that address the challenges of the 21st century.
8.1. Biopolymers: Sustainable Alternatives to Traditional Polymers
Biopolymers are polymers derived from renewable resources, such as plants and microorganisms. They are biodegradable and compostable, making them a sustainable alternative to traditional polymers derived from fossil fuels. Biopolymers are used in packaging, agriculture, and medicine.
8.2. High-Performance Polymers: Materials for Demanding Applications
High-performance polymers are materials that exhibit exceptional strength, heat resistance, and chemical resistance. They are used in demanding applications, such as aerospace, automotive, and electronics. High-performance polymers are often synthesized using advanced polymerization techniques and contain specialized monomers.
8.3. Smart Polymers: Responding to the Environment
Smart polymers are materials that respond to changes in their environment, such as temperature, pH, or light. They can change their shape, color, or properties in response to these stimuli. Smart polymers are used in a wide range of applications, including drug delivery, sensors, and actuators.
9. Frequently Asked Questions About Polymers (FAQ)
| Question | Answer |
|---|---|
| What is the difference between a polymer and a plastic? | Plastic is a type of polymer that can be molded into different shapes. Not all polymers are plastics. |
| Are all polymers synthetic? | No, many polymers are natural, such as proteins, carbohydrates, and nucleic acids. |
| Are polymers harmful to the environment? | Some polymers, particularly plastics, can be harmful to the environment if not properly disposed of. However, polymer recycling and the development of biopolymers are helping to address these concerns. |
| What are some examples of biopolymers? | Examples of biopolymers include cellulose, starch, chitin, and polylactic acid (PLA). |
| What are the benefits of using polymers? | Polymers are versatile, durable, lightweight, and relatively inexpensive to produce. They can be tailored to have a wide range of properties, making them suitable for countless applications. |
| How are polymers made? | Polymers are made through a process called polymerization, where small molecules called monomers are joined together to form long chains. There are two main types of polymerization: addition polymerization and condensation polymerization. |
| What is cross-linking in polymers? | Cross-linking is the process of forming chemical bonds between polymer chains. This increases the strength, elasticity, and heat resistance of the polymer. |
| What are some applications of polymers in medicine? | Polymers are used in medical implants, drug delivery systems, wound dressings, and tissue engineering. |
| How can I learn more about polymers? | Visit WHAT.EDU.VN to explore in-depth articles, resources, and answers to all your questions about polymers and other scientific concepts. Feel free to ask any questions you have on our platform for free answers. |
| What is the role of polymers in everyday life? | Polymers are integral to everyday life, present in clothing, packaging, electronics, vehicles, and construction materials, providing benefits like durability, flexibility, and protection. |

10. Ask Your Questions on WHAT.EDU.VN
Still have questions about what polymers are, their properties, or their applications? Don’t hesitate to ask! At WHAT.EDU.VN, we provide a free platform for you to ask any question and receive answers from experts and knowledgeable community members.
We understand that finding reliable information and getting your questions answered can be challenging and costly. That’s why we created WHAT.EDU.VN: to provide a free, accessible, and user-friendly platform for anyone to ask questions and get answers.
10.1. Why Choose WHAT.EDU.VN?
- Free: Ask any question and get answers without any cost.
- Fast: Receive quick and accurate responses from experts and community members.
- Easy to Use: Our platform is designed for simplicity and ease of navigation.
- Knowledgeable Community: Connect with a community of experts and enthusiasts.
- Comprehensive Information: Access a wealth of information on a wide range of topics.
10.2. How to Ask a Question on WHAT.EDU.VN
- Visit our website: WHAT.EDU.VN
- Create a free account or log in.
- Click on the “Ask a Question” button.
- Type your question clearly and concisely.
- Select the appropriate category for your question.
- Submit your question.
Our team of experts and community members will review your question and provide you with helpful and informative answers.
11. Contact Us
For any inquiries or assistance, please feel free to contact us:
- Address: 888 Question City Plaza, Seattle, WA 98101, United States
- WhatsApp: +1 (206) 555-7890
- Website: what.edu.vn
We are here to help you find the answers you need!
